|
|
Information box |
The main purpose of this site is to extend the
intraoperative monitoring of the pelvic organs as part of the
general nervous system to include the neurophysiologic
parameters with intraoperative navigation guided with Skyra 3
tesla MRI and other radiologic facilities to merge the
morphologic and histochemical data in concordance with the
functional data, trying during that to include the laparoscopic
surgeries with merging LION procedures in patients with spinal
cord injuries.
 CNS Clinic
CNS Clinic
Located in Jordan Amman near Al-Shmaisani hospital, where all
ambulatory activity is going on.
Contact: Tel: +96265677695, +96265677694.
 Skyra running
Skyra running
A magnetom Skyra 3 tesla MRI with all clinical applications
started to run in our hospital in 28-October-2013.
 Shmaisani hospital
Shmaisani hospital
The hospital where the project is located and running diagnostic
and surgical activity. |
|
 |
|
 |
 |
Intraoperative
Neurophysiological Monitoring of the Sacral
Nervous System |
 |
 |
ABSTRACT |
 |
The basic functional neuroanatomy of the genitourinary and
anorectal systems is briefly described. These systems are
involved in the so-called sacral functions (micturition,
defecation, erection, ejaculation, etc.). In this
section we describe clinical neurophysiological tests of the
functional integrity of the sacral neuromuscular system used for
diagnostic purposes. Later we deal with intraoperative
neurophysiological monitoring of the lumbosacral nervous system.
The validity of different intraoperative monitoring techniques
of this system is summarized.
 |
INTRODUCTION |
 |
The functions involving the genitourinary and anorectal systems
are uniquely
controlled by the complex interaction of the vegetative and the
somatic nervous
system. Insofar as it is the sympathetic, sacral parasympathetic and somatic
systems that constitute
the most important peripheral nervous structures controlling
these functions,
they may also be referred to as sacral functions. The functions
themselves
(micturition, defecation, erection, etc.) are now better
understood because with introduction of methods of measuring the different functional
parameters (urodynamics,
faecodynamics, measurements of the sexual response) that provide
for better diagnosis of dysfunction. The awareness that such
dysfunction is also
a consequence of damage to neural structures has also greatly
increased. On the
other hand, it has become possible to better define the various
lesions to the
nervous system by electrophysiological methods. However, these
methods by
and large document only the somatic sacral nervous system and
its central
pathways [1]. Nevertheless, such information is clinically
relevant because (1)
the somatic nervous system plays a part in all sacral functions,
and (2) the
somatic and parasympathetic sacral systems are closely related,
and information
on the somatic system may therefore be a relevant indicator of
the overall neurogenic
lesion in several clinical situations.
In fact, the common denominator of lower urinary tract,
anorectal, and
sexual functions is that many of their efferent and afferent
pathways travel at
least partly in close vicinity. They “share” common spinal cord
regulatory segments
(the upper lumbar segments—sympathetic efferents; the middle and
lower sacral segments—parasympathetic and somatic efferents).
Even the long
pathways connecting the relevant spinal cord segments with
higher levels of the
central nervous system are situated close together. Thus it is
not uncommon
that in lesions to the nervous system, and particularly in
lesions affecting the
spinal cord, cauda equina, sacral plexus, and pudendal nerve,
the sacral functions
are affected together. Dysfunctions may arise not only from
disease or
trauma, but also from inadvertent lesions to the previously
named structures
during invasive procedures, particularly several surgeries
involving the pelvic
organs and the spinal canal. The consequences of lesions to the
neurocontrol
of sacral functions include disturbing sensory phenomena like
pain, dysethesia,
urgency and frequency, loss of genital sensation, bladder or
rectal fullness,
and subsequent retention, obstipation, soiling, incontinence,
and erectile dysfunction.
Neurogenic damage may lead to lost coordination between detrusor
and sphincter function, leading to high bladder pressures and
upper urinary
tract dysfunction. Incontinence may—particularly in
motor-disabled persons—lead to problems with hygiene, skin problems, infections, and
decubiti.
Although neurogenic sexual dysfunction may not be
life-threatening, it can be
extremely disruptive psychologically and can lead to severe
emotional and
interpersonal problems. All these may be particularly tragic
when they are a
consequence of an inadvertent intraoperative lesion.
A whole array of clinical neurophysiological diagnostic methods
has been
modified for use in the anogenital area, including
electromyographic methods
and reflex, conduction, and evoked potential studies [2,3].
These methods are
routinely employed in uroneurological and neuro-urological
laboratories for
diagnostics and follow-up in patients with (suspected)
neurogenic sacral dysfunction.
Trials were performed to establish some of
these neurophysiological
methods in the operating room, to help the surgeon identify
particular sacral nervous structures, and to monitor the
function of the sacral
neuromuscular system during surgery [4–7].
2 FUNCTIONAL ANATOMY
Although the functional anatomy of the genitourinary and
anogenital systems is highly complex, it need not be considered in detail because
only the gross
anatomy of the relevant somatic nervous structures can be
approached by clinical
neurophysiological methods that are applicable in the operating
room environment.
Most of the information relevant for intraoperative
neurophysiology
can be summarized as follows.
Afferent fibers from the mucosa and skin of the genitoperineal
region travel
mostly with the pudendal nerves. The distally most accessible
group of sensory
fibers are the dorsal nerves of the penis (or clitoris). The
sensory fibers from the
genital, perineal, and anal region enter through the dorsal
spinal roots S2–S5
into the spinal cord and synapse (through interneurons) with
sphincteric motor
neurons. The afferent information also ascends (the primary
sensory neurons
synapsing to higher-order sensory neurons at various levels) via
the spinothalamic
and dorsal column tracts, the lemniscal system and thalamocortical tracts,
and finally to the somatosensory cortex (at its interhemispheric
location) [8].
The sphincteric lower motor neurons in the midventral spinal
grey matter of
the second to fourth sacral spinal cord segments (the “Onuf
nucleus”) are under
voluntary control from the motor cortex. Somatic motor nerve
fibers leave
through the ventral roots and the sacral plexus, combining into
the pudendal
nerves; direct branches innervate the levator ani and the anal
sphincter [8]. The
external urethral sphincter may be innervated by sacral somatic
fibers traveling
via splanchnic nerves [9] or the pudendal nerve [10], or
possibly both.
2.1 NEURAL CONTROL OF THE LOWER
URINARY TRACT
The lower urinary tract (LUT) is innervated by three sets of
peripheral nerves.
The pelvic parasympathetic nerves arise at the sacral level of
the spinal cord
(they excite the bladder and relax the urethra). The sympathetic
nerves arise
from the upper lumbar segments and inhibit the bladder body,
modulate transmission
in bladder parasympathetic ganglia, and excite the bladder base
and
urethra. The somatic efferents and afferents from the S2–S4
sacral roots innervate
pelvic floor muscles (levator ani) both through direct branches
and by the
pudendal nerve, which innervates also the perineal muscles,
including the anal
and urethral sphincter.
All of these nerves contain both efferent and afferent nerve
fibers that are
controlled by centers in the brain and particularly important
centers in the
brainstem. Long tracts in the spinal cord subserve the
spinobulbospinal reflex
pathway, which is relevant for coordinated detrusor-sphincter
function and
normal micturition. The dorsal pontine tegmentum is established
as an essential
control center for micturition (with a close anatomical
relationship with the
locus coeruleus). While different types of sensation of the
lower urinary tract
travel both in the anterolateral and the dorsal part of the
spinal cord, the
descending (motor) pathways lie within the lateral aspects of
the spinal cord.
2.2 ANORECTUM
Touch, pin-pricks, and hot and cold stimuli can be perceived in
the anal canal
to a level of up to 15 mm above the anal valves. The epithelium
in the area from
about 10–15 mm above the valves has a rich sensory nerve supply
made up of
both free and organized nerve endings. The sensory endings in
the hairy perianal
skin are similar to those in hairy skin elsewhere. The afferent
nerve pathway
for anal canal sensation is by the inferior hemorrhoidal
branches of the
pudendal nerve. Sensory pathways from the rectum and the bladder
travel in
the pelvic visceral nerves to the sacral cord, but some afferent
information is
probably also related to hypogastric nerves entering the spinal
cord at the thoracolumbar
level.
Functionally, the most important part of the smooth musculature
of the
anorectum is the internal anal sphincter, which is responsible
for about 85% of
the resting pressure in the lumen of the canal. The smooth
musculature of rectal
walls (and of the detrusor) receives extrinsic motor innervation
from the sacral
parasympathetic outflow arising in the intermediolateral cell
columns of sacral
cord segments S2–S4. These first-order neurons send axons that
emerge with
the ventral spinal nerve roots to synapse with second-order
neurons lying
within the pelvic plexus or the visceral walls. The sympathetic
nerve supply
arises from the thoracolumbar chain and travels in the
hypogastric nerve to
innervate visceral smooth muscle directly, and also via a
modulatory influence
on parasympathetic function at the level of the pelvic plexus.
The internal anal
sphincter is probably controlled both by sympathetic (hypogastric)
and sacral
parasympathetic pathways, but the inhibition brought about by
rectal distention
(the important rectoanal inhibitory reflex) is predominantly an
intramural
one.
The external anal sphincter is innervated by the pudendal nerve
and
occasionally also by a perineal branch of S4. The neurons of the
sphincter
motor nucleus (Onuf’s nucleus) are under voluntary control via
corticospinal
pathways.
Normal defecation is probably triggered by filling of the rectum
from the sigmoid
colon, and the signals from stretch receptors in the rectal wall
and pelvic
floor muscles are interpreted at the conscious level as a desire
to defecate. The
extension of the rectum causes reflex relaxation of the smooth
internal sphincter
muscle. Voluntary relaxation of the striated sphincter muscle
permits defecation,
which is assisted by colonic pressure waves and abdominal
straining. If
defecation is to be deferred, brief conscious contraction of the
voluntary sphincter
allows time for recovery of internal sphincter tone and
relaxation of the
rectum to accommodate filling. Conscious appreciation of the
desire to defecate
and intentional control over defecation are conferred by
suprasacral neural
influences. The precise way in which the autonomic, pyramidal,
extrapyramidal,
and sensory pathways integrate to achieve a reliable and
predictable anorectal
function is not yet fully understood [11].
2.3 SEXUAL ORGANS
Of the sexual functions affected by neurogenic lesions, research
has centered on
the male functions, and particularly on erection. Erection can
be initiated in the
brain and/or follow genital stimulation; in sexual activity a
combination of both
is probably involved.
Neurogenic erectile dysfunction due to peripheral lesions can be
secondary
to the disruption of sensory nerves contributing to the afferent
arm of reflex
erection or to the disruption of autonomic nerves that mediate
arterial dilatation
and trabecular smooth muscle relaxation. Erectile dysfunction
can occur
from disruption of the relevant pathways in centers within the
spinal cord (both
suprasacral and sacral), cauda equina, the sacral plexus, the
pelvic plexus, the
cavernosal nerves, and the pudendal nerves. Particular pelvic
surgeries such as
radical prostatectomy or cystoprostatectomy lead to a high
percentage of
mostly neurogenic erectile dysfunction; the lesion occurs in the
pelvic plexus
or in the cavernosal nerves located in the posterolateral aspect
of the prostate.
Ejaculation can be abolished by a lesion to the sympathetic
innervation of
the bladder neck (leading to a retrograde ejaculation) and by
disruption of the
sensory and (particularly) motor nerves innervating the perineal
muscles,
whose contraction leads to expulsion of the semen. It can also
be abolished by
central lesions.
A disturbed sexual response in females is due to (1) afferent
lesions leading
to loss of sensitivity of the perineal area, and (2) efferent
lesions leading to a loss
of lubrication, loss of clitoral erection, and pelvic floor
muscle denervation.
3 CLINICAL NEUROPHYSIOLOGICAL TESTS
IN DIAGNOSTICS
Since the function of all the aforementioned systems relies on
neural control,
clinical neurophysiological tests have been introduced to
support and supplement
clinical evaluation in patients. The tests comprise
electrophysiologic
methods of testing conduction through motor and sensory pathways
(both
peripheral and central) and electromyographic methods.
Traditionally, in testing
both the lower urinary tract and anorectal function, the EMG
signal
obtained from sphincter muscles has been used to delineate the
sphincter activity
patterns in relationship to micturition or defecation. In
addition to that,
electromyographic methods have been used to distinguish between
normal and
neuropathic pelvic floor muscles. Conduction tests have been
introduced to
evaluate the integrity of different reflex pathways (sacral
reflexes), the individual
motor pathways (pudendal nerve terminal latency, MEP), and
sensory pathways
(penile sensory neurography, SEP). In addition, autonomic tests
have also
been introduced (sympathetic skin response, corpus cavernosum
EMG). For
diagnostic purposes a single testing is performed without
knowledge of the previous
status of the investigated structure. In this diagnostic
situation, results
have to be compared to values obtained from healthy subjects.
The tests of conduction
have been found to be relatively insensitive to axonal lesions
because
amplitudes of responses vary widely in the control population
(particularly due
to technical reasons), and conduction may remain normal in
partial lesions.
Thus, in the diagnostic situation, the ability of the concentric
needle EMG to
detect abnormal spontaneous activity as an indicator of
denervation, and
changes of motor unit potentials as indications of
reinnervation, has been found
to be particularly helpful. EMGs and recordings of the
bulbocavernosus reflex
(indicating the potency of the lower sacral reflex arc) have
been proposed as the
basic battery of tests for evaluation of patients with sacral
dysfunctions and suspected
neurogenic involvement [12]. From conduction tests, only
recordings
of the sacral reflex and SEP after dorsal penile or clitoral
nerve stimulation have
been suggested since they have been validated by extensive
clinical studies.
They may be of value in selected patients with suspected
peripheral (i.e., bulbocavernosus
reflex testing) and central nervous system (i.e., SEP testing)
lesions [3, 13]. The other neurophysiological tests have been
suggested as
useful in further research. The corpus cavernosum EMG is the
most controversial
of the tests so far described. It is not yet well clarified
whether the signal
really originates from penile smooth muscle; validation of the
method would
offer a most important source of information on penile
innervation status,
which is necessary for erection.
4 INTRAOPERATIVE
CLINICAL NEUROPHYSIOLOGY
With appropriate
modifications of methods
it is technically feasible to record in anesthetized patients
(a) dorsal root action
potentials (DRAPs) after pudendal nerve stimulation, (b)
pudendal somatosensory
evoked potentials over the conus, the spinal cord, and the
scalp, (c)
sphincter muscle EMG responses to sacral ventral root
stimulation and motor
cortex stimulation, and (d) the bulbocavernosus reflex. Only one
of the aforementioned
techniques has been used extensively enough to gather pertinent
information regarding the practical relevance of sacral nervous
system monitoring
during surgical interventions. However, the techniques are
expected to
be valuable safeguards against inadvertent lesioning of nervous
structures that
would lead to some (neurogenic) dysfunction of micturition,
defecation, or the
sexual response. Further studies are required to clarify these
issues.
4.1 BASIC TECHNICAL ASPECTS OF STIMULATION
FOR INTRAOPERATIVE SACRAL MONITORING
In order to obtain bioelectrical signals useful for monitoring
purposes in the different
segments of the sacral neuromuscular system, it is necessary to
depolarize
the nervous system at particular segments. Up to now only
electrical
stimulation has been appropriate for this purpose. Stimulation
can be applied
to either the sensory part or the motor part of the system
(afferent versus efferent
events; Fig. 9.1). At present, most intraoperative monitoring of
the sacral
system has relied on responses evoked from stimulation of the
sensory system,
apart from recording of anal sphincter muscle responses upon
stimulation of
ventral spinal roots or the motor cortex.
|
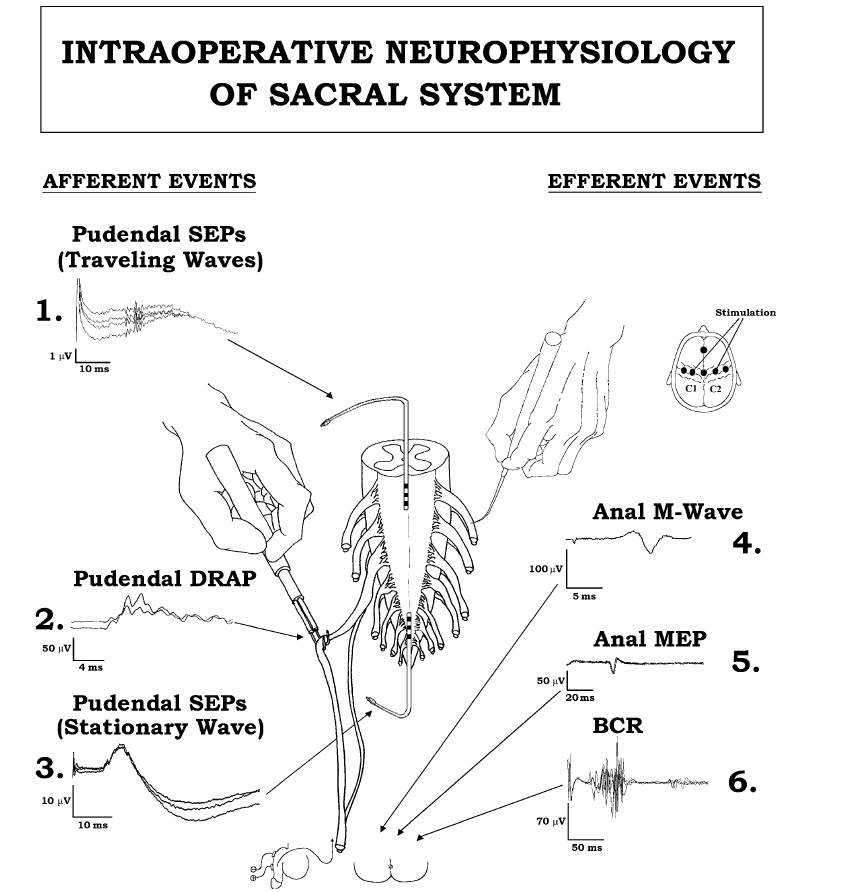 |
 |
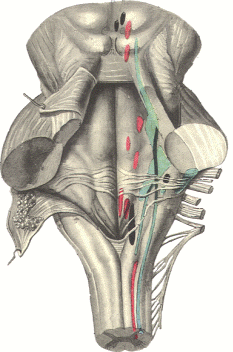
| FIGURE 9.1 Neurophysiological events used to intraoperatively
monitor the sacral nervous
system. Left, “afferent” events after stimulation of the dorsal
penile or clitoral nerves and recording
over the spinal cord: (1) pudendal SEPs, traveling waves, (2)
pudendal DRAPs, and (3) pudendal
SEPs, stationary waves, recorded over the conus. Right,
“efferent” events: (4) anal M wave recorded
from the anal sphincter after stimulation of the S1–S3 ventral
roots, (5) anal motor-evoked potentials
recorded from the anal sphincter after transcranial electrical
stimulation of the motor cortex,
and (6) bulbocavernosus reflex obtained from the anal sphincter
muscle after electrical stimulation
of the dorsal penile or clitoral nerves. Reprinted from Deletis,
V. (2001). Neuromonitoring. In “Pediatric
neurosurgery,” 4th ed. (D. MacLeone, ed.), pp. 1204–1213. W.B.
Saunders, Philadelphia. |
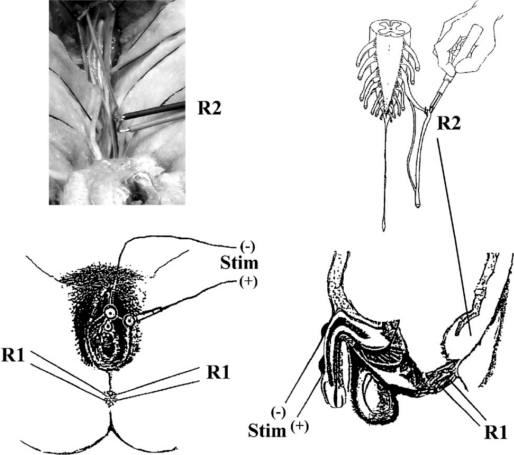
FIGURE 9.2 Lower left, position of the electrodes over the
clitoris and labia majora for the stimulation
of the dorsal clitoral nerves. Lower right, position of the
electrode for stimulating the dorsal
penile nerves. R1 = recording BCR from anal sphincter. Upper
right, schematics of recording DRAPs
with a hand-held hook electrode (R2) over the exposed dorsal
sacral roots of the cauda equina.
Upper left, intraoperative picture of DRAP recordings. |
The appropriate peripheral sensory structures that are available
for stimulating
purposes are the two adjacent dorsal penile or clitoral nerves.
These
nerves are stimulated by silver/silver chloride cup EEG-type
electrodes (EEG
electrodes) placed on the dorsal surface of the penis or
clitoris (this electrode
representing the cathode). The other electrode (anode) is placed
either distally
on the penis (1–2 cm apart from the proximal electrode) or on
the adjacent
labia (Fig. 9.2). In small children with short penises, the
anode may be attached
on the ventral side of the penis. The dorsal side of the penis
must be scrubbed
gently with Nuprep (D. O. Weaver & Co., Aurora, CO) before
placing electrodes
in order to avoid stimulus artifacts. The electrodes are filled
with electrode
cream and secured appropriately (Tegaderm; Smith and Nephew
Medical Limited,
Hull, England). The electrode sites are then bandaged with a few
layers of
gauze to prevent them from being displaced when the patient is
moved onto the
operating table. Electrode impedances should be kept below 5 kΩ.
As a general rule, stimuli of 20 mA intensity at 0.2 ms duration
have been
delivered in procedures involving stimulation of the penis or
clitoris (at various
frequencies for different measurements, up to 13.3 Hz).
Stimulation can also be performed at the level of the spinal
roots; a handheld
sterile monopolar electrode can be placed under the appropriate
roots (or
rootlets, after the root is freed from neighboring roots and
lifted outside the
spinal canal). Square wave pulses of 1 to 2 mA intensity and 0.2
ms duration
are delivered (Fig. 9.1, right).
4.2 BASIC TECHNICAL ASPECTS OF RECORDING FOR
INTRAOPERATIVE SACRAL MONITORING
Bioelectrical activity from the sacral
neuromuscular system has
up to now been recorded from the sacral dorsal spinal roots, the
spinal cord, the
somatosensory cortex, and the anal sphincter muscle.
Recordings from dorsal spinal roots (dorsal root action
potentials, DRAPs)
are obtained by hand-held sterile bipolar hook electrodes (after
the root is freed
from neighboring roots and lifted outside the spinal canal). In
this case, the
electrode closer to the point of stimulation is the G1 (active)
electrode. Epoch
lengths of 0 to 50 ms are used for these recordings (Figs. 9.1,
9.2).
Recordings of pudendal spinal somatosensory evoked responses (SSEPs,
stationary
wave) are obtained by a spinal epidural electrode placed over
the conus
(S2–S4). These potentials are generated by interneurons of the
grey matter
within the S2–S4 segments of the spinal cord. Typically 100
responses are averaged
together; epoch lengths of 0–50 ms are used (Fig. 9.1).
Recordings of pudendal spinal somatosensory evoked responses (SSEPs,
traveling
wave) are obtained by a spinal epidural electrode inserted
anywhere over
the dorsal column of the spinal cord. These potentials are
generated by pudendal
afferents traveling within the dorsal columns. Typically 100
responses are
averaged together; epoch lengths of 0–50 ms are used (Fig. 9.1).
To obtain pudendal cerebral somatosensory evoked responses (CSEPs),
Screwtype
recording electrodes are placed on the scalp 2 cm behind CZ (G1
or active
electrode) and at FZ (G2 or reference electrode), according to
the somatotopic
representation in the primary somatosensory cortex related to
the International
10–20 System of scalp electrode placement. The active electrode
is placed in the
midline because the sacral segments are represented deep within
the medial
longitudinal interhemispheric fissure. For CSEPs, 100–200 traces
are typically
averaged together. Epoch lengths of 0–200 ms are used.
To record anal sphincter (EMG) responses, either surface-type
electrodes or
hook wire electrodes can be used. Given the close anatomical
relationship
between the small sphincter muscle and neighboring larger muscle
groups,
recording must be selective (e.g., with intramuscular hook wire
electrodes) if
the stimulation technique is “nonselective” (such as in the case
of ventral root
stimulation, as a consequence of which neighboring muscles are
also excited).
When the stimulation procedure is more specific (e.g., in
bulbocavernosus or
pudendoanal reflex monitoring), the recording may be obtained
with properly
attached surface-type electrodes.
Sterile hook wire recording electrodes (Teflon-coated, 76 μm
diameter wire
with a 3 mm bare tip) are introduced into the left and right
sides of the external
anal sphincter with sterile needles; these are immediately
removed carefully
from the sphincter (the hooked wires remaining in place). The
integrity of the
electrodes can be tested by passing a short train of 50 Hz
current at 10 mA and
observing sphincter contraction (if the patient is not paralyzed
at the time of
electrode placement). The electrode impedances of these
electrodes should be
checked, although clean recordings are usually still possible
with high electrode
impedances.
The epoch length used for anal sphincter EMG recordings varies
according
to the type of response; either single or few averaged responses
can be obtained
upon stimulation (of course, the patient should not be under the
influence of a
muscle relaxant during this procedure).
4.3 SPECIFIC SACRAL NEUROMUSCULAR SYSTEM
MONITORING PROCEDURES
4.3.1 Pudendal Dorsal Root Action Potentials (DRAPs)
In the treatment of spasticity (e.g., in cerebral palsy), the
sacral roots are
increasingly being included during rhizotomy procedures.
Lang [14] demonstrated that children who underwent L2–S2
rhizotomies had
an 81% greater reduction in plantar/flexor spasticity compared
to children
who underwent only L2–S1 rhizotomies. But, as more sacral dorsal
roots have
been included in rhizotomies, neurosurgeons have experienced an
increased
rate of postoperative complications, especially with regards to
bowel and bladder
functions.
In order to spare sacral function, we attempted to identify
those sacral dorsal
roots that were carrying afferents from pudendal nerves. To do
this, we used
recordings of dorsal root action potentials (DRAPs). Patients
were anesthetized
with isoflurane, nitrous oxide, fentanyl, and a short-acting
muscle relaxant
introduced only at the time of intubation. The cauda equina was
exposed
through a T12–S2 laminotomy or laminectomy and the sacral roots
were identified
using bony anatomy. The dorsal roots were separated from the
ventral
ones, and DRAPs were recorded by a hand-held sterile bipolar
hooked electrode
(the root being lifted outside the spinal canal) (Fig. 9.2). The
DRAPs were
evoked by electrical stimulation of the penile or clitoral
nerves. One hundred
responses were averaged together and filtered between 1.5 and
2100 Hz. Each
average response was repeated to assess its reliability.
Afferent activity from the
right and left dorsal roots of S1, S2, and S3 was always
recorded, along with
occasional recordings from the S4–S5 dorsal roots. DRAP
recording was successfully
obtained in the majority of patients, the
DRAPs were present in the S2 and S3 roots bilaterally in 19
patients, whereas
in 7 patients DRAPs were also present in both S1 roots (in 8
patients they were
present in the S1 root unilaterally). However, the response in
the S1 root was
never larger than the S2 or S3 root responses. The range of
amplitudes was
2.9–18.3 μV in S1 roots, 3.2–129.9 μV in S2 roots, and 4.6–333
μV in S3 roots.
Of special relevance was the finding that in 7.6% of these
children, all afferent
activity was carried by only one S2 root (Fig. 9.3, C and F).
These findings were
confirmed by a later analysis of the results of mapping in 114
children (72 male,
42 female, mean age 3.8 years) [5]. Mapping was successful in
105 out of 114
patients; S1 roots contributed 4.0%, S2 roots 60.5%, and S3
roots 35.5% of the
overall pudendal afferent activity. The distribution of
responses was asymmetrical
in 56% of the patients (Fig. 9.3, B, C, and F). Pudendal
afferent distribution
was confined to a single level in 18% (Fig. 9.3, A), and even to
a
single root in 7.6% of patients (Fig. 9.3, C and F). Fifty-six
percent of the
pathologically responding S2 roots during rhizotomy testing were
preserved
because of the significant afferent activity, as demonstrated
during pudendal
mapping. None of the 105 patients developed long-term bowel or
bladder
complications.
All our results in the early series of dorsal root mapping with
19 patients [4]
have been confirmed by analysis of the larger series of 105
patients [5]. With
this series we showed that selective S2 rhizotomy can be
performed safely without
an associated increase in residual spasticity, while at the same
time bowel
and bladder function are preserved by performing pudendal
afferent mapping [14].
Therefore, we suggest that the mapping of pudendal afferents in
the dorsal roots
should be employed whenever these roots are considered for
rhizotomy in children
with cerebral palsy without urinary retention. Preoperative
neurourological
investigation of the children should help in making appropriate
decisions;
for example, in children with cerebral palsy with hyperreflexive
detrusor dysfunction,
sacral rhizotomy may be considered to alleviate the problem. In
any
case, intraoperative mapping of sacral afferents should make
selective surgical
approaches possible and provide the maximal benefit for children
with cerebral
palsy.
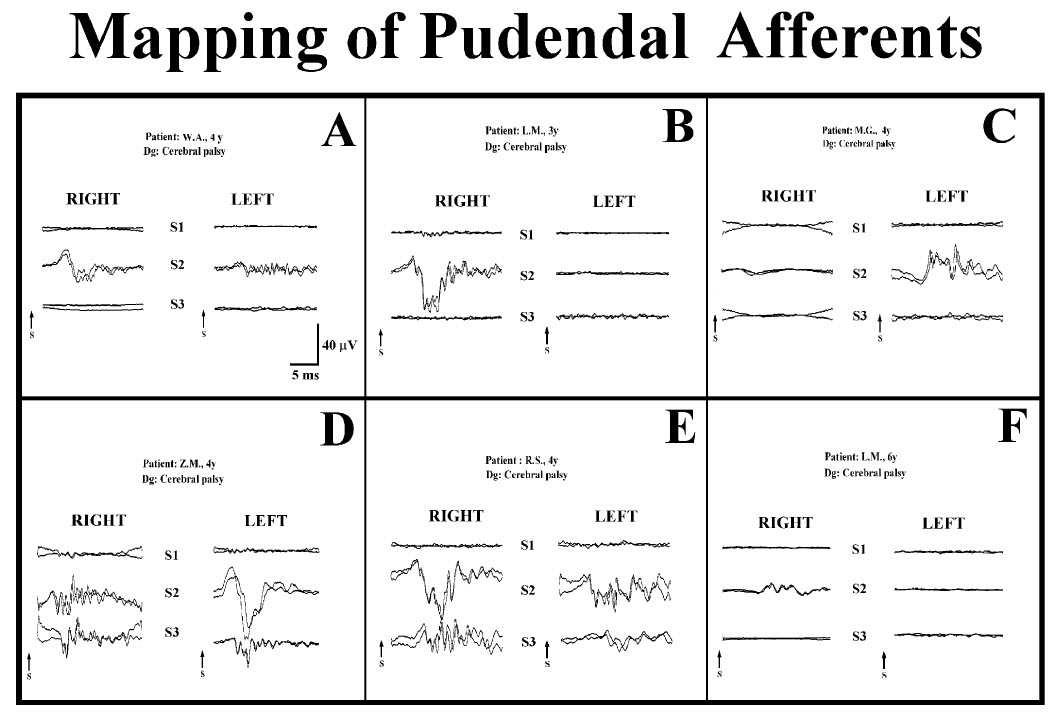 |
|
FIGURE 9.3 Six characteristic examples of DRAPs showing the
entry of a variety of pudendal nerve fibers to the spinal cord
via S1–S3 sacral roots. (A)
Symmetrical distribution of DRAPs confined to one level (S2) or
three levels (D). Asymmetrical distribution of DRAPS confined to
the right side (B), only
one root (C or F), or all roots except right S1 (E). Recordings
were obtained after electrical stimulation of bilateral
penile/clitoral nerves. |
Our results so far support the hypothesis that the root
distribution of afferent
fibers that are important for the control of micturition may be
similar to the
distribution of mucocutaneous afferent fibers from the pudendal
nerve. Only
further studies will clarify the complex functional anatomic
issues involved.
Recordings were performed in four children of both sexes, 2.5 to
7.0 years of
age. Small-amplitude (up to 1 μV) SSEPs, which were very stable,
could be
recorded with subdurally placed electrodes over the thoracic
spinal cord (traveling
waves), while a stationary wave could be recorded with a much
higher
amplitude (up to 10 μV) over the conus region. The latencies of
the spinal SEP
over the conus region ranged from 6.0 to 10.4 ms (Fig. 9.1,
left). The recordings
were made as a pilot study, and thus far only demonstrate the
ability to
obtain such recordings intraoperatively. Further employment of
this technique
showed that traveling waves are difficult to record, and
successful recording of
a stationary wave necessitates that the electrode be placed
strictly over the S2–S4
sections of the spinal cord.
4.3.3 Pudendal Cerebral Somatosensory Evoked Potentials (CSEPs)
Well-formed cerebral SEPs with amplitudes of 0.5–0.7 μV were
recorded on
dorsal penile nerve stimulation throughout spinal neurosurgical
procedures in
two adult male patients, 50 and 78 years of age. Stable P40
peaks were obtained.
The recordings were made as a pilot study and thus far only
demonstrate the
ability to obtain such recordings intraoperatively. Further
employment of this
method showed that this potential is very sensitive to
anesthetics; a formal feasibility
study has not yet been performed.
4.3.4 Anal Sphincter Motor Response Monitoring
In five children of both sexes, 2.5 to 9.0 years old, anal
sphincter muscle EMG
responses were recorded by stimulation of the ventral spinal
roots (L5, S1, S2,
S3, and S4) to identify ventral roots carrying motor fibers to
the sphincter
muscle. Recordings were obtained by surface conductive rubber
electrodes
(applied para-anally) and intramuscular hooked wire electrodes.
In surface
recordings, no unilateral responses could be identified, and
responses were
also obtained on stimulation of the L5 and S1 roots (on
stimulation of L5 and
S1 roots no adequate responses could be discerned from
simultaneous recordings
from intramuscular electrodes). The surface recorded responses
were recognized
as “nonspecific” (derived from neighboring muscles, most
probably
glutei [15]).
The latency of surface recorded responses was, as a rule,
shorter than the
latency of responses obtained from intramuscular electrodes,
which was between
5 and 8 ms. On electrical stimulation of the motor cortex, anal
sphincter
responses were recorded in a large group of anesthetized
patients without pyramidal
involvement. Because of polysynaptic connections of the
corticospinal
tract to the α-motoneuron of S2–S4, these responses are
moderately sensitive
to anesthetics (Fig. 9.1).
4.3.5 Bulbocavernosus Reflex (BCR) Monitoring
Intraoperative recordings were first performed in 15
neurosurgical patients (11
males, 4 females, 2–6 years old). Patients were without sacral
dysfunction and
were anesthetized with fenatyl and propofol or nitrous oxide
without the
influence of a muscle relaxant. Recordings from the anal
sphincter were
obtained by hooked wire electrodes and were recorded as a single
response.
Very reproducible responses could be obtained on double-pulse
stimulation,
the optimum interstimulus interval being found to be 3 ms and
the optimum
stimulation rate 2.3 Hz. Continuous periods of stimulation and
recording for
up to 10 min were repeatedly performed with very reproducible
results (Fig.
9.4). The reflex response was suppressed by the administration
of isoflurane
and nitrous oxide and was completely abolished by muscle
relaxants [16]. After
this pilot study, 119 patients were tested, 38 of which
underwent surgery without
risk and 81 of which underwent surgery with risk of damage to
sacral
structures. In all, 51 adults (19 to 64 years old, 32 male and
19 female) and
68 children (24 days to 17 years old, 30 male and 38 female)
took part in the
study. Patients were anesthetized with propofol, fenatyl, or
nitrous oxide
with a short-acting relaxant. Clinically, most patients had mild
to moderate
upper motor neuron deficits in the lower extremities, and no
patient had major
urinary problems. In all patients it was possible to record
reproducible reflex
response with the previously described method. In patients
without risk to the
sacral system, only a few minutes of the responses were recorded
to test their
feasibility, whereas in the patients at risk, continuous
monitoring was conducted.
|
 |
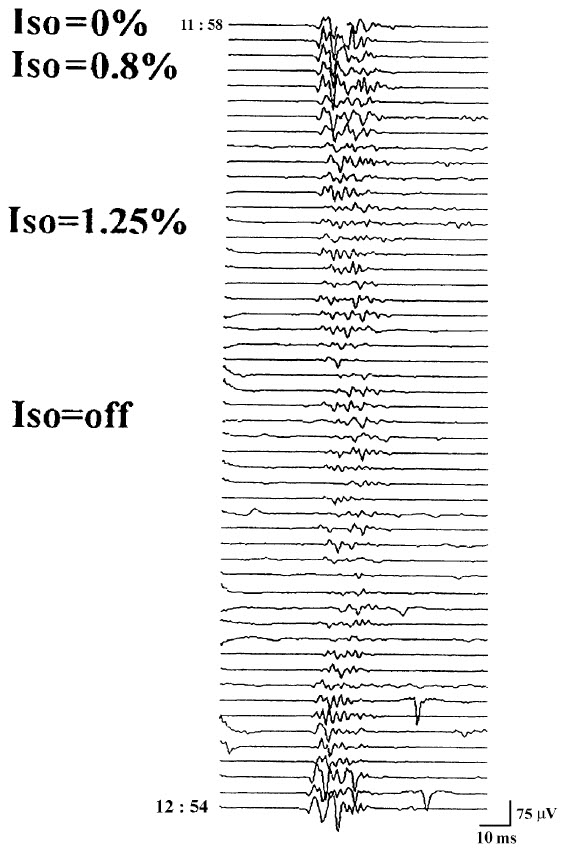 |
FIGURE 9.4 Continuous monitoring of the BCR for a period of 10
min showing the stability of
the BCR’s appearance. |
FIGURE 9.5 The influence of isoflurane (Iso) on the BCR. Note
that the response was almost completely
blocked when the concentration of 1.25% was administered for 15
min and did not recover
until almost 30 min after the isoflurane was discontinued.
Reprinted from [6]. |
The influence of volatile anesthetic was tested in 6 patients.
In 3
patients, BCR was suppressed by the administration of 1.25%
isoflurane for 15 min
(Fig. 9.5). Administration of nitrous oxide (60% inspired
concentration) for
15 min reduced the BCR in 3 patients, and the response was
completely abolished
by muscle relaxant in 2 patients [6].
Preliminary results conducted in 50 patients with a lumbosacral
tethered
cord showed no clear correlation between BCR and postoperative
sphincter control. The authors concluded that the complexity of
sphincter
innervation and segmental or suprasegmental control probably
accounts for this
discrepancy [17].
Up to now, close to 250 patients have been monitored using this
method. In
the last 100 patients, a train of four consecutive stimuli [7]
was used rather than
a double stimulus, providing even more robust responses. Some
problems have
been encountered in female patients in whom the stimulation
method is not as
robust as required; this problem needs particular attention from
the technician.
5 DISCUSSION AND CONCLUSIONS
The impetus to include intraoperative monitoring of sacral
nervous structures
came from concerns over optimizing care for children with
cerebral palsy.
These were successfully treated for their spasticity by
performing selective
dorsal rhizotomy; surgeons have always sought to minimize the
side-effects of
this procedure while maintaining its benefits (benefits being a
reduction in
muscle hypertonia, the side-effects being a partial or complete
loss of sensory
modalities). The first sacral dorsal roots have always been a
target for rhizotomy
in this procedure, but the S2 dorsal roots have become
increasingly considered
for rhizotomy. Not to treat the S2 dorsal roots is to leave
potentially abnormal
reflexive circuits that will continue to drive spasticity in the
musculature of the
leg. For this reason, the lesion zone was extended to include S2
dorsal roots,
and the result was a greater reduction in spasticity as compared
with children
in whom the lesion was extended only to the S1 segment.
Unfortunately, the
extension to include S2 roots was also associated with disorders
in micturition.
In one group of patients, before DRAP mapping was introduced,
24% experienced
urinary retention (which was only transient in most children).
Although
most of these children had both the left and right S2 roots cut,
2 of the children
had only one S2 root cut, and they also experienced retention.
The selective
dorsal rhizotomy procedure should be performed in young children
who are
certainly too young to assess sexual function; they are even too
young to be
completely confident that all symptomatic complaints regarding
micturition or
defecation and perineal sensation were being relayed. It was
this concern over
the preservation of genitourinary afferent function that led us
to develop the
technique of intraoperative neurophysiological identification of
the sacral roots
responsible for perineal sensation. In the first 31 children,
neurophysiological
identification of roots and rootlets carrying afferent activity
from the penile or
clitoral nerves led to zero micturition disturbances and allowed
for rhizotomy
of S2 roots or rootlets not carrying such afferent activity.
Therefore, maximum
possible antispastic effects could be achieved. The particularly
important lesson
we learned by doing very systematic recordings in S1–S2 and S3
roots bilaterally
(and in some children also S4 and S5 roots) is that although
most of our
patients showed evidence of pudendal afferents in S2 and S3
roots bilaterally,
about half of them also showed evidence of some afferent
activity in S1 (either
unilaterally or bilaterally). In slightly more than half of our
patients, the root
potentials were symmetrical, but the pudendal afferents of many
were irregularly
distributed across the sacral roots. In a few of the children
the afferentactivity was confined to a single root (either S2 or S3 root).
Since this may be
the most important afferent contribution from the genitourinary
area, even if
only the S2 dorsal roots are checked for relevant afferent
activity, they should
not be sacrificed if they show any such activity. The finding of
asymmetrical distribution
of fibers that may be confined to a single root is consistent
with the
work of Junemann et al. [18], who found that the majority of
motor fibers for
the urethral sphincter are carried by a single variable lower
sacral root. Therefore,
for some patients undergoing an operation in the area of the
cauda equina,
the sacrifice of one single root may have dire functional
consequences.
For other neurosurgical procedures in the lumbosacral spinal
canal (e.g.,
the release of a tethered cord or the removal of a tumor), other
authors have
proposed, and in a small series have performed, the
identification of motor and
sensory nerve roots. This has been achieved through continuous
monitoring of
electromyographic activity in muscles innervated by lumbosacral
segments, and
through monitoring of tibial nerve somatosensory evoked
potentials [19]. In
addition, monitoring pudendal SEPs should provide very relevant
complementary
information, and, as we have demonstrated, should not be
technically
demanding if the structures are preserved preoperatively.
Kothbauer et al. [19]
claim that intraoperative recordings saved operating time by
allowing the surgeon
more rapid and decisive preparation than would be possible on an
anatomical basis alone, and they also gave the impression that
the procedures
were safer. The recordings of spontaneous anal sphincter
electromyographic
activity during such operations have been described earlier
[20]. To accomplish
the two goals of neurophysiology (i.e., first, immediate
identification of structures
as functional nervous tissue and their distinction from other
tissue; and
second, continuous monitoring of the function of the relevant
nervous structures),
a battery of methods needs to be applied, and a whole set of
structures
needs to be assessed bilaterally. Therefore, both the lower
sacral segments and
sphincter muscles, and also the upper sacral segments and the
lumbar segments,
need to be included. Also, all these segments and several
functional
modalities need to be monitored more or less simultaneously. The
appropriate
setup for each surgical situation would need to be selected on
the basis of
anatomical and physiological considerations, and a compromise
between the
possible and the necessary would be sought.
Other authors have claimed that continuous EMG recording of
bursts or
trains of motor unit potentials or repetitive neurotonic
discharges elicited by
injury to the peripheral motor fibers have correlated with
postoperative transient
or permanent neurological deficit [21, 22] in the area of facial
nerves. The predictive
value of these “manipulation-evoked” discharges is only based on
empirical
data, but has been proposed to also be of value in the
lumbosacral segments
[19]. The electrophysiological identification of motor nerves is
already an integral
part of cranial nerve surgery [22] and should also provide a
similar service
in the region of the cauda equina.
Up to now, only recordings of DRAPs have been made in a large
number of
subjects (to identify sacral roots carrying genitourinary
afferents), and the electrophysiological
procedure decreased postoperative voiding disturbances [4, 5].
We propose, however, that the other intraoperative
electrophysiological recordings
of the sacral neuromuscular system that have been described
(spinal SEP,
CMAP of sphincter muscles, bulbocavernosus reflex recordings)
should prove
relevant in surgeries involving the sacral roots, the cauda
equina, and the conus
and should aid the surgeon in preventing inadvertent damage to
these structures.
These other procedures have not, however, been performed in an
adequate
number of patients, and their relevance cannot yet be appraised.
The
most interesting question is whether monitoring of the
bulbocavernosus reflex
for conus and cauda equina surgery could, as we are proposing,
replace and
even improve upon separate monitoring of motor and sensory
fibers of the
lower sacral roots.
As for surgeries involving the spinal cord above the conus, the
procedures
of pudendal SSEPs, cortical SEPs, and (possibly) motor evoked
potentials of the
anal sphincter muscle might be interesting in some selected
patient groups.
These would include groups in whom the preservation of sacral
function may
be particularly important (for instance, in scoliosis surgery in
patients with
advanced neuromuscular diseases who have heavily compromised
motor function
but no sacral dysfunction).
In other contexts, authors have argued that recordings from the
anal sphincter
cannot be taken as completely adequate information on the
functional status
of the urethral sphincter [18]. Since innervation of both
sphincters originates
from the same sacral segments (which also provide innervation
for the detrusor),
the monitoring of sphincter ani should generally mirror the
function of
relevant urethral structures.
As previously stated, the relatively limited experience with the
monitoring
of sacral structures cannot as yet prove that surgeries
accompanied by such
monitoring are easier and safer. For the time being, there is
anecdotal evidence
that difficult surgical decisions that have relied on the
results of intraoperative
neurophysiological measurements have not resulted in unexpected
neurological
deficits. Although the techniques described clearly provide the
surgeon with
additional information about nerve location and function, the
value of these
techniques must be further defined and documented. As Daube [23]
suggests,
it will be necessary to demonstrate that these techniques indeed
save
operative time, save anesthesia time, or—most
importantly—improve outcomes.
It will be difficult to demonstrate this without doing a
randomized study
of patients who are undergoing surgery with and without such
monitoring
techniques.
|
The main purpose of
intraoperative neurophysiologic monitoring is to reduce
postoperative neurological deficits, but more recently it has
become apparent that intraoperative recording of sensory evoked
potentials and electromyographic (EMG) potentials can also aid
the surgeon during many operations. The use of intraoperative
monitoring of sensory evoked potentials and EMG potentials to
reduce permanent postoperative deficits is based on the
assumption that changes in recordable electrical responses occur
as a result of injury, and that the injury is still reversible
at the time of detection if proper surgical intervention occurs.
Monitoring
of brain stem auditory evoked potentials (BAEPs) during
operations in which the vestibulocochlear nerve (cranial nerve
VIII) may be manipulated is now widespread, and the use of
monitoring to reduce the incidence of hearing loss due to
surgical manipulation of the vestibulocochlear nerve is steadily
increasing. However, intraoperative monitoring of visual evoked
potentials (VEPS), has not gained similar acceptance, mainly
because of the technical problems involved in generating a
suitable stimulus. Intraoperative recording of somatosensory
evoked potentials (SSEPs) is valuable during aneurysm surgery
and during other vascular operations, as well as during the
removal of tumors in which the tumor itself or the surgical
manipulation used to remove the tumor can be expected to affect
brain structures that are involved in the somatosensory system
and thus affect the generation of SSEPs.
The most
frequent use of intraoperative monitoring of SSEPs is in
operations involving the spinal cord, in which such monitoring
is now well established. More recently, intraoperative
transcranial stimulation of the motor cortex, by using either a
high-voltage electrical stimulation or a strong magnetic field
impulse in connection with recording of EMG potentials from the
motor system, has been introduced to reduce intraoperative
injuries to the spinal cord.
The
recording of EMG responses from muscles innervated by different
cranial nerves has been shown to be valuable in identifying
cranial motor nerves during operations to remove tumors when the
anatomy has been altered by the tumor or by previous operations;
this is particularly true for operations to remove acoustic
tumors and it has more recently been shown to be of value also
in operations to remove large tumors of the skull base. It has
been possible in a few types of operations to use
electrophysiologic methods to guide the surgeon in the operation
and to ensure before the operation has ended that the
therapeutic goal of the operation has been achieved.
The
objective of intraoperative monitoring of evoked potentials
(BAEPs, SSEPs. and VEPs) when used for the purpose of reducing
permanent postoperative neurological deficits is to detect
changes that occur during the operation. This differs from the
goal of the use of evoked potentials for diagnostic purposes, in
which a deviation from a normal value is of interest. In
intraoperative monitoring, normal values are of little interest;
instead, it is important to obtain a baseline recording from an
individual patient and then to compare the potentials that are
recorded during the operation to that baseline recording. Such a
baseline can usually be obtained after the patient has been
anesthetized but before the operation has begun.
|
 |
 |
Preservation
of the Facial Nerve During Operations in the
Cerebellopontine Angle |
 |
Monitoring of contractions of the facial muscles is
performed during operations in the ccrebellopontine
angle to help the surgeon locate the facial nerve
(CN
VII) when it is
not identifiable visually. Possibly just as important,
this technique makes it possible to determine which
portions of an acoustic tumor do not contain any part of
the facial nerve. This thus allows the surgeon to remove
portions of a tumor without risk of injuring the facial
nerve. Currently most such monitoring involves the
surgeon using a handheld stimulating electrode, which
carries short pulses of electrical current, to probe the
surgical field to identify the facial nerve. Various
methods are used to record the subsequent contractions
of the facial musculature. The facial muscle
contractions that are elicited by irritation and
manipulation of the facial nerve are just as important
as the contractions elicited by electrical stimulation
of the facial nerve for the purpose of assessing
injuries to the facial nerve.
Earlier, it
was customary to have an assistant observe any movement of the
face and then to communicate that fact to the surgeon. More
recently, the recording of EMG potentials from the facial
musculature and the recording of movements of the
face using electronic sensors have also been used to verity that the facial
nerve has been stimulated. The use of EMG recordings makes it
possible to assess the degree of facial muscle contraction
quantitatively, which was not possible when facial muscle
activity was assessed by visual observation of movements of the
face. The capability of making the recorded EMG potentials
audible allows the surgeon to hear when the facial nerve has
been stimulated and thus the surgeon has no need to rely on
communication with an assistant. For this purpose,
only a conventional audio amplifier and loudspeakers need to be
connected to the output of the EMG amplifier. Some commercial
equipment makes use of amplitude-sensing devices that elicit a
tone signal when the EMG potentials reach a preset amplitude.
However, making the (original) EMG signal audible is
advantageous because it provides valuable information that is
lost when such amplitudesensing devices are used. Although the
audible EMG signal provides most of the information that is
needed when monitoring the facial nerve, an oscillographic
display of the recorded EMG potentials is also advantageous, in
that it allows assessment of the amplitudes and latencies of
the recorded EMG potentials. When the movements of the face are
recorded by electronic sensors, the movements can be made to
elicit a sound (horn), but an oscillographic display of the
electrical signals produced by movement detectors has limited
value.
|
Because the
facial nerve is often split into several fascicles when a large
acoustic tumor has displaced it, it is important that recordings
be made from the entire face. If only portions of the facial
musculature (e.g., the lower face or the upper face) are
monitored, failure to locate all parts of the facial nerve could
result in inadvertent removal of or injury to portions of the
facial nerve: this would result in postoperative paralysis of
part of the face. Usually the record EMG activity is performed between
two
electrodes, one placed
on the forehead and one on the lower face (Fig.-1A), when
monitoring facial function intraoperatively. In addition to
recording activity from all muscles on the side of the face on
which the electrodes have been placed, this particular
arrangement of recording electrodes will also record
contractions of the masseter and temporal muscles. These muscles
are innervated by the motor portion of the trigeminal nerve (portio
minor), and there is the possible risk of mistaking the fifth
(motor) nerve for the seventh nerve when probing the cerebellopontine angle for the facial nerve in operations to
remove large acoustic tumors that have progressed rostral to the
trigeminal nerve. The EMG response to stimulation of the
trigeminal nerve, recorded in the way illustrated in Fig-1A,
however, can easily be distinguished from the response to
stimulation of the facial nerve, because the latency of the
recorded EMG signal differs in the two situations [1.5 ms and 5
to 6 ms. respectively,(Fig-1B). An alternative way
to distinguish between the
response to facial nerve stimulation and that to stimulation of
the motor portion of the trigeminal nerve
(CN
V) is to record from the
masseter muscle using a pair of electrodes connected to a
separate differential EMG amplifier (Fig-1A). This
recording will almost exclusively yield the response of the
masseter muscle, and thus is a good indicator of stimulation of
the motor portion of the trigeminal nerve. |
 |
|
Fig-1: A-Electrode
placement for recording EMG potentials
from facial muscles and a separate recording of the
response from the masseter muscle. B- EMG potentials
recorded from electrodes, placed as shown in A,
elicited by electrical stimulation of the trigeminal
and facial nerves in the CPA. C- Hand held
stimulating electrodes for intracranial localization
of the cranial nerves. |
There are
several advantages to using EMG recordings as a measure of
muscle contraction rather than using a single sensor that
records movements of the face. First, EMG potentials from
practically all of the facial muscles can be recorded on a
single channel [Fig-1A),
whereas several sensors are needed to cover the
entire face when movements are being recorded. Second.
recording EMG potentials makes it possible to measure the
latencies of the responses accurately, which enables one to
differentiate between activation of the trigeminal nerve and
activation of the facial nerve (Fig-1B). Third, the amplitude of
the EMG response is roughly a measure of how many
nerve fibers are functioning. and it therefore provides
valuable quantitative information about the degree of injury to
the facial nerve.
There are
essentially two ways that electrical stimulation can be applied
to the facial nerve: one is by using a bipolar stimulating
electrode, and the other is by using a monopolar electrode.
the difference being that the bipolar electrode has a higher
degree of spatial specificity. However, because it is only the
negative phase of the stimulus that is effective, only one of
the two prongs of a bipolar stimulus electrode will stimulate
the facial nerve effectively. Therefore, the orientation of a
bipolar electrode is important. A monopolar. hand-held,
stimulating electrode does not have these disadvantages and
although it is less selective than a bipolar stimulating
electrode it is preferable for intraoperative use (Fig-1C).
A monopolar stimulating electrode is easy to use and its
stimulating power is not affected by its orientation, as is the
case for a bipolar stimulating electrode. By using a monopolar,
handheld, stimulating electrode and having EMG potentials made
audible, the surgeon can probe a large area of a tumor quickly
and "map" the tumor to locate portions of the tumor where no
part of the facial nerve is present so that it can be removed
safely. The use of this technique can often reduce
considerably the time required to remove the tumor because
large portions of the tumor can be removed without risk of
causing injury to the facial nerve. Later during removal of
the tumor, when it becomes important to identify the facial
nerve accurately so that injury to the nerve can be avoided, the
same monopolar stimulator can be used and the area of tissue
that it stimulates can be varied by varying the voltage that is
applied through the electrode.
The
electrical stimulation should consist of negative (rectangular)
impulses of short duration (0.1 to 0.2 ms), and the stimulus
strength should be no greater than necessary to produce a
contraction. Some older types of stimulators make use of large
current and some even make use of direct current. Such
stimulators should not be used because of poor specificity and,
particularly, because of the risk of injuring the nervous tissue
with the electrical current used to stimulate the facial nerve.
Because
shunting of electrical current from the stimulating probe can
vary widely when the surgical field is wet compared to when it
is relatively dry, it is advantageous to use a relatively
constant-voltage stimulator rather than the more conventional
constant-current type of stimulator.
Constant-current stimulation in connection with the use of a
"flush tip" stimulating electrode (i.e., an electrode that is
insulated all the way to its tip) has been advocated by some
investigators. When constant-voltage stimulation is used, the
same amount of stimulus current will flow through a specific
tissue (e.g., the facial nerve), regardless of how much shunting
occurs. If constant-current stimulation is used, the same total
current is delivered, but the amount of current that passes
through a specific volume of tissue depends greatly on how much
current is shunted away; in this case the stimulus strength
depends heavily on whether the field is wet or dry.
When a
facial nerve stimulator is used to identify regions of a tumor
where no part of the facial nerve is present, the stimulus
strength should be set so that it will activate the facial nerve
if the nerve is within a small distance of the tip of a
monopolar stimulating electrodes.
Audible
monitoring (by means of a loudspeaker) of the EMG activity of
the facial muscles that is evoked by mechanical stimulation of
the facial nerve provides valuable feedback to the surgeon
during the delicate resection of portions of an acoustic tumor
that involve the facial nerve. Such continuous
monitoring of facial EMG activity (without electrical
stimulation) is of great value, particularly when the surgeon is
removing a large tumor, parts of which may be firmly adherent to
the facial nerve. If the facial nerve is being heated by electrocoagulation, or heated by the drilling of bone adjacent
to the facial nerve, transient or sustained facial muscle
activity will result. Although such monitoring of facial
EMG activity is important in reducing the risk of permanent
damage to the facial nerve, it must be pointed out that the
facial nerve can be injured permanently from surgical
manipulation without any EMG response being noted; thus, injury
from sharp dissection will most likely not result in recordable
EMG activity (or any movement of the face). For this reason,
when the surgeon is dissecting near the facial nerve spontaneous
EMG activity should not be relied upon for assurance that the
facial nerve remains intact; in this situation, electrical
stimulation of the facial nerve should be used frequently to
identify the facial nerve so that the surgeon remains aware at
all times of the exact location of the facial nerve.
 |
Monitoring of
the Extraocular Nerves during Skull Base Surgery |
 |
|
In
operations to remove tumors of the skull base, several cranial
motor nerves are often in the operative field and are thus at
risk of injury from surgical manipulation. This is particularly
true in operations within the cavernous sinus, where the nerves
that innervate the extraocular muscles [the oculomotor (CN
III), trochlear (CN IV), and abducens (CN VI) nerves] may be
difficult to identify visually or may be displaced by the tumor.
Recording EMG potentials from the extraocular muscles while the
surgical field is being probed with a hand-held stimulating
electrode similar to that used for stimulating the facial nerve
(Fig-1C) can aid the surgeon in locating the respective
nerves. EMG potentials can easily be recorded from these
muscles via needle electrodes inserted percutaneously into the
respective muscle (Fig-2). The potentials recorded from the extraocular muscles in response to electrical stimulation of the
respective nerves are easy to distinguish (Fig-3A).
More
recently, surface electrodes for recording EMG activity from the
extraocular muscles have been developed. Facial nerve function
should also be monitored in these operations, using the methods
just described. Continuous recording of EMG potentials from
these muscles is also important, because injury to the respective nerves from mechanical manipulation and from heat
during electrocoagulation will often result in transient or
sustained EMG activity, as was described for the facial nerve.
Thus, such activity can be an important aid in preserving these
nerves. |
 |
 |
 |
|
Fig-2: A-Electrode
placement for recording from facial,
extraocular muscles and the tongue. The
reference electrodes are all placed on the
forehead on the opposite side, to avoid
recording from the facial muscles at the
same time. B-Electrode placement with
intraoperative recordings from extraocular
and facial muscles. BAEPs and VEPs included. |
Fig-3: A- EMG potentials
recorded from the extraocular and facial
muscles in response to intracranial
electrical stimulation of the respective
cranial nerves, using the electrode
arrangement as seen in Fig-2. B-EMG
potentials from the tongue in response to
stimulation of the hypoglossal nerve. |
 |
Monitoring of
Other Cranial Motor Nerves |
 |
Identification of the hypoglossal nerve (CN XII) can be
facilitated by recording EMG potentials from the tongue (Figs-2A and
3B), and monitoring of the accessory nerve (CN XI) can
conveniently be done by placing pairs of needle EMG electrodes
in the trapezius muscle (Fig-2A). The motor portions of
the glossopharyngeal (CN IX) and vagus (CN
X) nerves can be monitored by
stimulating the respective nerves electrically and recording EMG
activity from the muscles that these nerves innervate in a way
similar to that just described for the facial nerve and the
nerves that innervate the extraocular muscles. A pair of
needle electrodes placed in the soft palate will record the EMG
response to stimulation of the glossopharyngeal nerve and
electrodes placed in the supraglottic region are suitable for
recording of the EMG response from laryngeal muscles that are
innervated by the recurrent nerve of the vagus nerve. Electrodes
placed on the endotracheal tube can record surface EMG
potentials from laryngeal muscles. A balloon placed on the tip of an endotracheal tube
as a pressure recording device has been used to record
contractions of the laryngeal muscles for monitoring of the
recurrent laryngeal nerve. This technique could be useful in
monitoring the more central portions of the vagus nerve.
One should
be aware when monitoring the glossopharyngeal, vagus, and
accessory nerves using electrical stimulation that there are
risks involved. For example, a supramaxial stimulation of the
accessory nerve may result in so strong a muscle contraction
that dislocation of joints or physical injury to muscles and
tendons may result. Electrical stimulation of the
glossopharyngeal and vagus nerves may cause cardiovascular
effects and should therefore be done cautiously.
 |
Monitoring of
Facial Nerve Function during Microvascular
Decompression for Hemifacial Spasm |
 |
Microvascular decompression to relieve hemifacial spasm is one
of only a few operations in which intraoperative
neurophysiologic monitoring can aid the surgeon in achieving the
therapeutic goal of the operation. It is generally
accepted that hemifacial spasm is caused by vascular compression
of the facial nerve as it exits the brain stem. and that
microvascular decompression of the root exit zone of the facial
nerve is the most effective treatment of this
disorder. However, it is not always obvious from
an exploration of the root exit zone of the facial nerve which
of several vessels is causing the spasm, and a certain (small)
number of patients who have undergone this operation have
experienced spasm postoperatively. Some of these patients had to
be reoperated upon, depending on the severity of the spasm.
 |
 |
|
Fig-4A: Electrode placement for
recording the abnormal muscle response in hemifacial
spasm. |
Fig-4B: The abnormal muscle
response recorded from the mentalis muscle to
electrical stimulation of the temporalis branch. The
left record is before opening the dura, showing
variable EMG activity in addition to component with
a latency of 10 ms. After decompression, the low
amplitude spontaneous activity is indicative for
slight facial nerve injury. |
In studies of the pathophysiology of hemifacial spasm, it was found that
an abnormal muscle response that seems to be characteristic of
the disorder disappears instantaneously when the offending
blood vessel is moved off the intracranial portion of the facial
nerve (Fig-4). This abnormal muscle response, which has
a latency of about 10 ms is seen when one branch of the
facial nerve is stimulated electrically and recordings are made
from muscles that are innervated by other branches of the facial nerve. By monitoring this abnormal muscle response intraoperatively, it is possible to identify the offending blood
vessel and to ensure that the nerve has been fully decompressed
by watching for the cessation of the abnormal response (Fig.4B) When using this method
it was found that even veins can cause hemifacial spasm and
that in many cases there was more than one blood vessel
compressing the facial nerve. There are reasons to assume that
at least some of the patients who experienced only partial
relief from their symptoms before this type of monitoring was
introduced did so because more than one vessel was affecting the
facial nerve and only one of the offending vessels was moved off
the facial nerve during the operation.
 |
Monitoring of
Brain Stem Auditory Evoked Potentials |
 |
|
Intraoperative monitoring of BAEPs is commonly performed to
reduce the risk of hearing loss as a result of intraoperative
manipulation of the vestibulocochlear nerve in operations in
the cerebellopontine angle. This
is important in operations on acoustic tumors, in which hearing
preservation is anticipated, as well as in microvascular
decompression operations on cranial nerves and in other
operations in the cerebellopontine angle.
BAEPs are
commonly recorded between electrodes placed on the vertex and on
the earlobe (or mastoid) of the ear to which the sound is
applied. BAEPs are best elicited by click sounds presented at a
rate of 20 to 40 pulses/s (pps) at intensities of 100 to 110
peak equivalent sound pressure (Pe SPL), The normal BAEPs are
characterized by 5 to 7 vertex-positive peaks that are generated
as the different structures of the ascending auditory pathway
are successively activated (Fig.-5).
Because the
BAEPs are generated by fiber tracts and nuclei of the ascending
auditory pathway in the brain stem, the vestibulocochlear nerve,
the cochlear nuclei, and the lateral lemniscus being the most
important generators of BAEPS-recording of these
potentials is not only useful for detecting injury to the
vestibulocochlear nerve but may also be of value in operations
in which the brain stem is being manipulated or when circulation
to the brain stem may be compromised. The nuclei of
the ascending auditory pathway are sensitive to manipulations of
the brain stem, and there
are indications that BAEPs may be more
sensitive
in detecting such effects than
are changes in heart rate and blood pressure. A change in the latency of peak V while that
of peak III remains unchanged (increased III - V interpeak
latency) indicates an effect from surgical manipulation on
structures located in the region of the superior olivary complex
on either side or on the lateral lemniscus and its nucleus on
the side contralateral to the side on which the BAEPs are being
elicited (tumor side) (Fig-6). It is therefore more valuable
to monitor BAEPs during other operations in which manipulation
of the brain stem may occur, such as the removal of large
acoustic tumors or other tumors of the cerebellopontine angle.
|
 |
|
Fig-5: Neural generators
of BAEPs. DCN: dorsal cochlear nucleus, VCN:
ventral cochlear nucleus. SO: superior
olivary complex. LL: lateral lemniscus. IC:
inferior colliculus. MG: medial geniculate. |
 |
|
Fig-6:
Changes of latencies and amplitudes of peaks
III and V of BAEPs as a function of time
during surgery. BAEPs were elicited by
stimulating the opposite ear relative
to the tumor location. |
 |
Monitoring of
Somatosensory Evoked Potentials |
 |
|
SSEPs are
important in monitoring sensory conduction in the spinal cord. SSEPs elicited by stimulation of the median
nerve and recorded from the contralateral parietal region of the
scalp (C3 or C5) using a noncephalic reference are characterized
by a series of positive and negative peaks (Fig-7). P9, P11,
and PI4 are short-latency, positive peaks that are generated at
the level of the brachial plexus (P9). spinal entry (P11). and
termination of the dorsal column in the dorsal column nuclei
(PI4). The bilateral N18 is generated in brain stem
nuclei, such as the superior colliculus. whereas the
contralateral N20 is assumed to be generated in the primary
cortex. The waveform of the SSEPs and the presence of certain
components depend on the recording sites (Fig-7). For
monitoring the spinal cord, SSEPs are elicited by electrical
stimulation of sensory nerves on the leg (peroneal or posterior
tibial nerve).
Only when the cervical spine is
considered is it appropriate to use SSEPs elicited by
stimulation of the upper limb (median nerve at the wrist). The
response to stimulation of the lower limb is recorded from
electrodes placed on the scalp, vertex to a midline front
reference or vertex to a noncephalic reference. The
different components appear with longer latencies than those of
SSEPs elicited from the median nerve. and the pattern of the
peaks is more complex (Fig-8). One reason that lower limb SSEPs
are more complex than those elicited from
the upper limbs is related to the fact that
ascending neural activity elicited by
stimulating lower limbs travels in two
separate fiber tracts in the spinal cord.
Only part of the sensory information is
relayed in the dorsal column nuclei (nucleus
gracilis).
|
 |
 |
|
Fig-7:
SSEPs in response to electrical stimulation
of the median nerve. A- The thick line is a
record from the contralateral parietal
region with a noncephalic reference. B-
Records from prerolandic region with a
noncephalic reference.
|
Fig-8:
SSEPs in response to stimulation of the left
posterior tibial nerve at the ankle.
A-Records from a midfrontal (pFz).
B- Record from midpaietal (Pz)
with a noncephalic reference on the
shoulder. C- Difference between A and B.
|
|
Neural activity that travels in the dorsal
column is mainly elicited by skin receptors. Fast-conducting
afferents that conduct activity elicited by stretch receptors,
group Ia and II afferents from muscle spindles, and tendon
organs travel in a spinothalamic pathway in the dorsal lateral
funiculus of the spinal cord and synapse in the nucleus
gracilis, after which they join other afferents in the medial
lemniscus traveling toward the thalamic relay nuclei. The slow
cutaneous afferents from the lower limbs that travel in the
dorsal column are relayed in the dorsal column nuclei (gracilis
nucleus). These afferents have a large range of conduction
velocities, which causes a temporal dispersion in the elicited
activity, which is the cause of the low amplitude of the early
response from stimulation of lower limbs. This makes the SSEPs
from lower limbs qualitatively different from the SSEPs elicited
by stimulation of upper limbs and, together with the longer
distance from the location of the stimulation to the brain stem
structures, explains why the far field SSEP responses to
electrical stimulation of lower limbs are less well defined than
the SSEPs elicited by stimulation of the upper limbs, where all
somatic afferents trawl in the dorsal column and are all relayed
in the dorsal column nuclei (cuneate nucleus). Although
recording using a noncephalic reference is appropriate for
identifying the neural generators of SSEPs, the unfavorable
signal-to-noise ratio in such recordings has made it more common
to place the reference electrode on the scalp when SSEPs are
used for intraoperative monitoring. This reduces the pick-up of
electrical interference signals. When the spinal cord is to be
monitored, all components of SSEPs that originate from
structures that are rostral to the location where injury may
occur can be utilized for detecting injuries.
However, late components of
the SSEPs are affected by anesthesia, and it would
therefore be advantageous to use early components
such as the P14 in upper limb SSEPs.
Unfortunately, the early
components of lower limb SSEPs are less well-defined and usually
cannot be used for intraoperative monitoring.
Because the
blood supply to the portion of the spinal cord that comprises
the ascending somatosensory pathway is different from that of
the descending motor tracts, there is a possibility that the
motor system can be injured without any noticeable change
occurring in SSEPs. Thus, it seems possible that severe injury
could go unnoticed when only SSEPs are monitored. Although it
has been disputed whether this is in fact a real risk, there is
a need to be able to monitor both motor and sensory systems
during operations in which the spinal cord is at risk. Although
traditional techniques can be used to monitor the somatosensory
system intraoperatively, there are considerable technical
obstacles in eliciting motor responses upon stimulation of the
motor cortex. Therefore, such monitoring of the descending motor
tracts is not done routinely. It has, however, been shown that
it is indeed possible to elicit motor responses by electrical
and magnetic transcranial stimulation of the cortex,
thus a prerequisite for developing routine methods for
intraoperative monitoring of motor tracts.
Intraoperative monitoring of SSEPs is valuable as an indicator
of decreased cerebral perfusion in regions of the brain on which
the generation of more long-latency components of the SSEPs
depend. The development of these methods, which are now in
routine use, had been pioneered by Symon et. al, who made use of recordings of SSEPs elicited by electrical
stimulation of the median nerve at the wrist. The use of this
method is based on the finding that there is a rather close
relationship between the disappearance of electrical activity
and a decrease in the cerebral blood flow to 15 to 18 ml/100 g
per min (or below), and there are changes in the late components
of the SSEPs that occur before the perfusion reaches these low
levels. SSEPs are therefore valuable in estimating changes in
cerebral blood flow.
The most
useful parameter of SSEPs as an indicator of a decrease in
cerebral blood flow is the central conduction time, which is
the time difference between the occurrence of the components of
the SSEPs that can be recorded at the neck (P14) and the N20
component that is recorded at
the contralateral parietal skull (C3-C5) (Fig-9). Central conduction time is not affected
by changes in peripheral neural conduction or by
age, and it has been shown
to correlate with cerebral blood flow when blood flow is reduced
below a certain value.
|
 |
|
Fig-9: Measures of
central conduction time from recordings of the SSEPs
that are elicited by electrical stimulation of the
median nerve at the wrist. All recordings were done
with a noncephalic reference. A- Parietal scalp. B-
Frontal scalp. C- Spinal C6 spine. |
 |
Monitoring of
Visual Evoked Potentials |
 |
During
operations in which the optic nerve or the optic tract is being
manipulated it would seem to be beneficial to monitor
VEPS. However, the results of such monitoring have been
generally disappointing because the changes in the recorded
potentials correlate poorly with postoperative changes in
vision. This is most likely due to limitations inherent in the
techniques currently available for intraoperative stimulation of
the visual system, and perhaps to inadequate knowledge of how to
interpret the changes in the VEPs that may occur during surgical
manipulations of the optic nerve or optic tract: however,
essentially there are no practical problems involved in
recording VEPs intraoperatively. At present, the only
practical type of visual stimulation that can be used intraoperatively is the "flash" type. It has been shown that
VEPs elicited by a changing pattern (pattern
reversal-checkerboard pattern) are much more useful
diagnostically than VEPs recorded in response to flash
stimulation, however, eliciting VEPs by pattern reversal
technique requires that a pattern be focused on the retina,
which is not possible to accomplish intraoperatively.
 |
Effects of
Anaesthesia |
 |
Although
recordings of BAEPs are not noticeably affected by any common
anesthetic regimen using inhalation anesthetics. barbiturates.
and other intravenous anesthetics, intraoperative
monitoring of cranial motor nerves cannot be done if the
patient is paralyzed, because such monitoring depends on
recording muscle responses by EMG technique. Components of the
upper limb SSEPs that occur with latencies longer than 16 ms are
usually affected by general surgical anesthesia.
The most
commonly used anesthetic regimen for neurosurgical operations
involves giving a strong narcotic, such as fentanyl for
analgesia together with an inhalation agent, such as nitrous
oxide, and small amounts of halogenated agents ("balanced
anesthesia"). Because the patient must be kept from moving,
he or she must be paralyzed when such a regimen is used. Normally
some form of a muscle endplate blocker
(pancuronium, vecuronium.
etc.) is used for this purpose. Therefore, balanced anesthesia
makes it impossible to record EMG potentials because of muscle
relaxation. However, intraoperative neurophysiologic monitoring
is fast becoming a regular component of the surgical arena, and
it is now common to adjust the anesthesia regimen to the
requirements of the specific type of monitoring to be done.
For instance, the requirement of being able to record EMG
potentials has resulted in the use of inhalation anesthesia
throughout the operation. possibly with the addition of a small
amount of a narcotic agent and benzodiazepines such as midazolam.
This type of anesthesia has been used for many
years without any noticeable difficulties or complications and
is now in common use when intraoperative monitoring of muscle
activity is performed. However, while such an anesthesia regimen
has no noticeable effect on recording EMG potentials or BAEPs
and short-latency SSEPs. it is likely to suppress later
components of SSEPs and it greatly suppresses motor evoked
potentials. Some anesthetic agents such a propofol,
that can be
administered intravenously have less suppressive effect on
cortical responses and are thus being used during operations in
which several different kinds of evoked potentials and EMG
responses are to be monitored.
 |
Preoperative
Assessment of Patients for Intraoperative
Monitoring |
 |
Patients in
whom intraoperative neurophysiologic monitoring of evoked
potentials is to be performed should have the systems that are
to be monitored quantitatively evaluated preoperatively. For
example, if a patient's BAEPs are to be monitored, his or her
hearing status should be evaluated preoperatively. Testing
should include at least a pure tone audiogram and a
determination of speech discrimination scores, and the patient's
ear canals should be checked for obstruction (from cerumen. etc.).
If the patient has no
hearing or if it is not possible to obtain an interpretable BAEP
before the operation, it will not be possible to obtain one
during the operation. If, on the other hand, an interpretable BAEP can be obtained before the operation but not
intraoperatively. the cause is most likely technical: such
problems then must be corrected. Similarly, if SSEPs are to be
recorded intraoperatively, a recording should be performed
preoperatively to ascertain that the patient has normal SSEPs or
to establish the degree of abnormality so that it can be taken
into consideration before the intraoperative recordings are
made. Factors such as neuropathies that may affect neural
conduction should be ruled out or assessed quantitatively, if
possible. If the extraocular museles are to be monitored, their
function should be evaluated preoperatively by
state-of-the-art methods. When facial EMG responses are to be
monitored, the function of the facial musculature should be
evaluated preoperatively. Preoperative evaluation of sensory and motor systems that may be at risk during an operation is also
essential, because this, in connection with similar tests done
postoperatively, the only way that deficits from the
operation can be assessed quantitatively.
 |
Determination
of Benefits of Intraoperative Neurophysiologic
Monitoring |
 |
Intraoperative neurophysiologic monitoring of evoked potentials
has been introduced for use in neurosurgical
operations for long time, and it is therefore natural that the method has
been viewed with skepticism in the early period of its
implementation, and suffered from a demand for proof of the
benefits. As with several other procedures allied
to such operations, it has generally been difficult to design
studies to provide quantitative estimates of the benefits in
terms of reduced postoperative complications. This is largely
because many surgeons believe that intraoperative monitoring is
valuable in reducing postoperative deficits and therefore will
not allow random selection of patients to be monitored. This
makes it impossible to study the efficacy of such techniques by
a double-blind methodology.
Comparing
complications before and after the introduction of
intraoperative monitoring thus seems to be the only practical
way to assess the value of intraoperative monitoring: however,
the results of such evaluations are influenced by any changes
in the operative technique introduced at or after the
institution of monitoring. Despite this complication.
comparisons have been made between the rate of complications
(such as facial palsy) of surgical procedures (such as acoustic
tumor removal) before and after the introduction of intracranial
monitoring of cranial nerve function. Such studies have shown a
significant decrease in complication rate after the introduction
of intraoperative monitoring.
It is
generally accepted now that intraoperative monitoring of facial
nerve function during operations on acoustic tumors is of value
in preserving the facial nerve. A study of a less frequently
performed operation, that to relieve hemifacial spasm, showed
that the efficacy of the operation was higher after
intraoperative monitoring was introduced. Further, the number
of patients who needed to be reoperated upon because of
recurrent or unrelieved spasm decreased from about 15 percent to
almost zero. Finally, the frequency of hearing loss as a
complication of microvascular decompression operations in the cerebellopontine angle decreased radically after the
introduction of intraoperative monitoring of BAEPs during such
operations.
Because
intraoperative neurophysiologic monitoring can many times
identify exactly which step in an operation that caused an
injury is likely to result in a permanent neurologic deficit, it
has contributed to the development of better and safer operating
techniques. When evaluating the benefits of intraoperative
neurophysiologic monitoring it must be pointed out that the
benefit from intraoperative neurophysiologic monitoring depends
on the level of experience of the surgeon. Thus a very
experienced surgeon will not have the same degree of benefit
from this technique as might a less experienced surgeon. There are several advantages of the use of
intraoperative neurophysiologic monitoring, but they are
difficult to measure quantitatively; nevertheless, they are
great enough to lead most surgeons who have operated with the
aid of such monitoring to demand that it continue.
|
 |
|