|
|
Information box |
The main purpose of this site is to extend the
intraoperative monitoring of the pelvic organs as part of the
general nervous system to include the neurophysiologic
parameters with intraoperative navigation guided with Skyra 3
tesla MRI and other radiologic facilities to merge the
morphologic and histochemical data in concordance with the
functional data, trying during that to include the laparoscopic
surgeries with merging LION procedures in patients with spinal
cord injuries.
 CNS Clinic
CNS Clinic
Located in Jordan Amman near Al-Shmaisani hospital, where all
ambulatory activity is going on.
Contact: Tel: +96265677695, +96265677694.
 Skyra running
Skyra running
A magnetom Skyra 3 tesla MRI with all clinical applications
started to run in our hospital in 28-October-2013.
 Shmaisani hospital
Shmaisani hospital
The hospital where the project is located and running diagnostic
and surgical activity. |
|
 |
|
 |
 |
PELVIC
NEUROANATOMY |
 |
 |
INTRODUCTION |
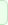 |
The sensory information about the pelvic organs
travels from the periphery to the central nervous system
(afferent fibers) and the motor fibers, either somatic
(voluntary red muscles) or sympathetic and parasympathetic
innervate the the glands, smooth involuntary white muscles. The
autonomic nerves are either sympathetic coming through the the
anterior roots of T1-L1, or parasympathetic coming through the
anterior roots of S2-5. In the inferior hypogastric plexus all
autonomic nerves are interconnected. The somatic innervation to
the pelvic organ running through the lumbar and sacral plexuses.
The hypogastric plexus and the sympathetic trunk are the
contributors for autonomic innervation to the pelvis.
 |
THE SACRAL
PLEXUS |
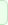 |
The sacral plexus is formed from the
lumbo-sacral trunk and ventral rami of S1 down to S5. Anatomical
variations are present in population with lumbarization or
sacralization of the area. The pelvic parasympathetic splanchnic
nerves arise from S2,3,4, which innervate the descending colon,
the rectum, urinary bladder and genital organs.
2.1 NEURAL CONTROL OF THE LOWER
URINARY TRACT
The lower urinary tract (LUT) is innervated by three sets of
peripheral nerves.
The pelvic parasympathetic nerves arise at the sacral level of
the spinal cord
(they excite the bladder and relax the urethra). The sympathetic
nerves arise
from the upper lumbar segments and inhibit the bladder body,
modulate transmission
in bladder parasympathetic ganglia, and excite the bladder base
and
urethra. The somatic efferents and afferents from the S2–S4
sacral roots innervate
pelvic floor muscles (levator ani) both through direct branches
and by the
pudendal nerve, which innervates also the perineal muscles,
including the anal
and urethral sphincter.
All of these nerves contain both efferent and afferent nerve
fibers that are
controlled by centers in the brain and particularly important
centers in the
brainstem. Long tracts in the spinal cord subserve the
spinobulbospinal reflex
pathway, which is relevant for coordinated detrusor-sphincter
function and
normal micturition. The dorsal pontine tegmentum is established
as an essential
control center for micturition (with a close anatomical
relationship with the
locus coeruleus). While different types of sensation of the
lower urinary tract
travel both in the anterolateral and the dorsal part of the
spinal cord, the
descending (motor) pathways lie within the lateral aspects of
the spinal cord.
2.2 ANORECTUM
Touch, pin-pricks, and hot and cold stimuli can be perceived in
the anal canal
to a level of up to 15 mm above the anal valves. The epithelium
in the area from
about 10–15 mm above the valves has a rich sensory nerve supply
made up of
both free and organized nerve endings. The sensory endings in
the hairy perianal
skin are similar to those in hairy skin elsewhere. The afferent
nerve pathway
for anal canal sensation is by the inferior hemorrhoidal
branches of the
pudendal nerve. Sensory pathways from the rectum and the bladder
travel in
the pelvic visceral nerves to the sacral cord, but some afferent
information is
probably also related to hypogastric nerves entering the spinal
cord at the thoracolumbar
level.
Functionally, the most important part of the smooth musculature
of the
anorectum is the internal anal sphincter, which is responsible
for about 85% of
the resting pressure in the lumen of the canal. The smooth
musculature of rectal
walls (and of the detrusor) receives extrinsic motor innervation
from the sacral
parasympathetic outflow arising in the intermediolateral cell
columns of sacral
cord segments S2–S4. These first-order neurons send axons that
emerge with
the ventral spinal nerve roots to synapse with second-order
neurons lying
within the pelvic plexus or the visceral walls. The sympathetic
nerve supply
arises from the thoracolumbar chain and travels in the
hypogastric nerve to
innervate visceral smooth muscle directly, and also via a
modulatory influence
on parasympathetic function at the level of the pelvic plexus.
The internal anal
sphincter is probably controlled both by sympathetic (hypogastric)
and sacral
parasympathetic pathways, but the inhibition brought about by
rectal distention
(the important rectoanal inhibitory reflex) is predominantly an
intramural
one.
The external anal sphincter is innervated by the pudendal nerve
and
occasionally also by a perineal branch of S4. The neurons of the
sphincter
motor nucleus (Onuf’s nucleus) are under voluntary control via
corticospinal
pathways.
Normal defecation is probably triggered by filling of the rectum
from the sigmoid
colon, and the signals from stretch receptors in the rectal wall
and pelvic
floor muscles are interpreted at the conscious level as a desire
to defecate. The
extension of the rectum causes reflex relaxation of the smooth
internal sphincter
muscle. Voluntary relaxation of the striated sphincter muscle
permits defecation,
which is assisted by colonic pressure waves and abdominal
straining. If
defecation is to be deferred, brief conscious contraction of the
voluntary sphincter
allows time for recovery of internal sphincter tone and
relaxation of the
rectum to accommodate filling. Conscious appreciation of the
desire to defecate
and intentional control over defecation are conferred by
suprasacral neural
influences. The precise way in which the autonomic, pyramidal,
extrapyramidal,
and sensory pathways integrate to achieve a reliable and
predictable anorectal
function is not yet fully understood [11].
2.3 SEXUAL ORGANS
Of the sexual functions affected by neurogenic lesions, research
has centered on
the male functions, and particularly on erection. Erection can
be initiated in the
brain and/or follow genital stimulation; in sexual activity a
combination of both
is probably involved.
Neurogenic erectile dysfunction due to peripheral lesions can be
secondary
to the disruption of sensory nerves contributing to the afferent
arm of reflex
erection or to the disruption of autonomic nerves that mediate
arterial dilatation
and trabecular smooth muscle relaxation. Erectile dysfunction
can occur
from disruption of the relevant pathways in centers within the
spinal cord (both
suprasacral and sacral), cauda equina, the sacral plexus, the
pelvic plexus, the
cavernosal nerves, and the pudendal nerves. Particular pelvic
surgeries such as
radical prostatectomy or cystoprostatectomy lead to a high
percentage of
mostly neurogenic erectile dysfunction; the lesion occurs in the
pelvic plexus
or in the cavernosal nerves located in the posterolateral aspect
of the prostate.
Ejaculation can be abolished by a lesion to the sympathetic
innervation of
the bladder neck (leading to a retrograde ejaculation) and by
disruption of the
sensory and (particularly) motor nerves innervating the perineal
muscles,
whose contraction leads to expulsion of the semen. It can also
be abolished by
central lesions.
A disturbed sexual response in females is due to (1) afferent
lesions leading
to loss of sensitivity of the perineal area, and (2) efferent
lesions leading to a loss
of lubrication, loss of clitoral erection, and pelvic floor
muscle denervation.
3 CLINICAL NEUROPHYSIOLOGICAL TESTS
IN DIAGNOSTICS
Since the function of all the aforementioned systems relies on
neural control,
clinical neurophysiological tests have been introduced to
support and supplement
clinical evaluation in patients. The tests comprise
electrophysiologic
methods of testing conduction through motor and sensory pathways
(both
peripheral and central) and electromyographic methods.
Traditionally, in testing
both the lower urinary tract and anorectal function, the EMG
signal
obtained from sphincter muscles has been used to delineate the
sphincter activity
patterns in relationship to micturition or defecation. In
addition to that,
electromyographic methods have been used to distinguish between
normal and
neuropathic pelvic floor muscles. Conduction tests have been
introduced to
evaluate the integrity of different reflex pathways (sacral
reflexes), the individual
motor pathways (pudendal nerve terminal latency, MEP), and
sensory pathways
(penile sensory neurography, SEP). In addition, autonomic tests
have also
been introduced (sympathetic skin response, corpus cavernosum
EMG). For
diagnostic purposes a single testing is performed without
knowledge of the previous
status of the investigated structure. In this diagnostic
situation, results
have to be compared to values obtained from healthy subjects.
The tests of conduction
have been found to be relatively insensitive to axonal lesions
because
amplitudes of responses vary widely in the control population
(particularly due
to technical reasons), and conduction may remain normal in
partial lesions.
Thus, in the diagnostic situation, the ability of the concentric
needle EMG to
detect abnormal spontaneous activity as an indicator of
denervation, and
changes of motor unit potentials as indications of
reinnervation, has been found
to be particularly helpful. EMGs and recordings of the
bulbocavernosus reflex
(indicating the potency of the lower sacral reflex arc) have
been proposed as the
basic battery of tests for evaluation of patients with sacral
dysfunctions and suspected
neurogenic involvement [12]. From conduction tests, only
recordings
of the sacral reflex and SEP after dorsal penile or clitoral
nerve stimulation have
been suggested since they have been validated by extensive
clinical studies.
They may be of value in selected patients with suspected
peripheral (i.e., bulbocavernosus
reflex testing) and central nervous system (i.e., SEP testing)
lesions [3, 13]. The other neurophysiological tests have been
suggested as
useful in further research. The corpus cavernosum EMG is the
most controversial
of the tests so far described. It is not yet well clarified
whether the signal
really originates from penile smooth muscle; validation of the
method would
offer a most important source of information on penile
innervation status,
which is necessary for erection.
4 INTRAOPERATIVE
CLINICAL NEUROPHYSIOLOGY
With appropriate
modifications of methods
it is technically feasible to record in anesthetized patients
(a) dorsal root action
potentials (DRAPs) after pudendal nerve stimulation, (b)
pudendal somatosensory
evoked potentials over the conus, the spinal cord, and the
scalp, (c)
sphincter muscle EMG responses to sacral ventral root
stimulation and motor
cortex stimulation, and (d) the bulbocavernosus reflex. Only one
of the aforementioned
techniques has been used extensively enough to gather pertinent
information regarding the practical relevance of sacral nervous
system monitoring
during surgical interventions. However, the techniques are
expected to
be valuable safeguards against inadvertent lesioning of nervous
structures that
would lead to some (neurogenic) dysfunction of micturition,
defecation, or the
sexual response. Further studies are required to clarify these
issues.
4.1 BASIC TECHNICAL ASPECTS OF STIMULATION
FOR INTRAOPERATIVE SACRAL MONITORING
In order to obtain bioelectrical signals useful for monitoring
purposes in the different
segments of the sacral neuromuscular system, it is necessary to
depolarize
the nervous system at particular segments. Up to now only
electrical
stimulation has been appropriate for this purpose. Stimulation
can be applied
to either the sensory part or the motor part of the system
(afferent versus efferent
events; Fig. 9.1). At present, most intraoperative monitoring of
the sacral
system has relied on responses evoked from stimulation of the
sensory system,
apart from recording of anal sphincter muscle responses upon
stimulation of
ventral spinal roots or the motor cortex.
|
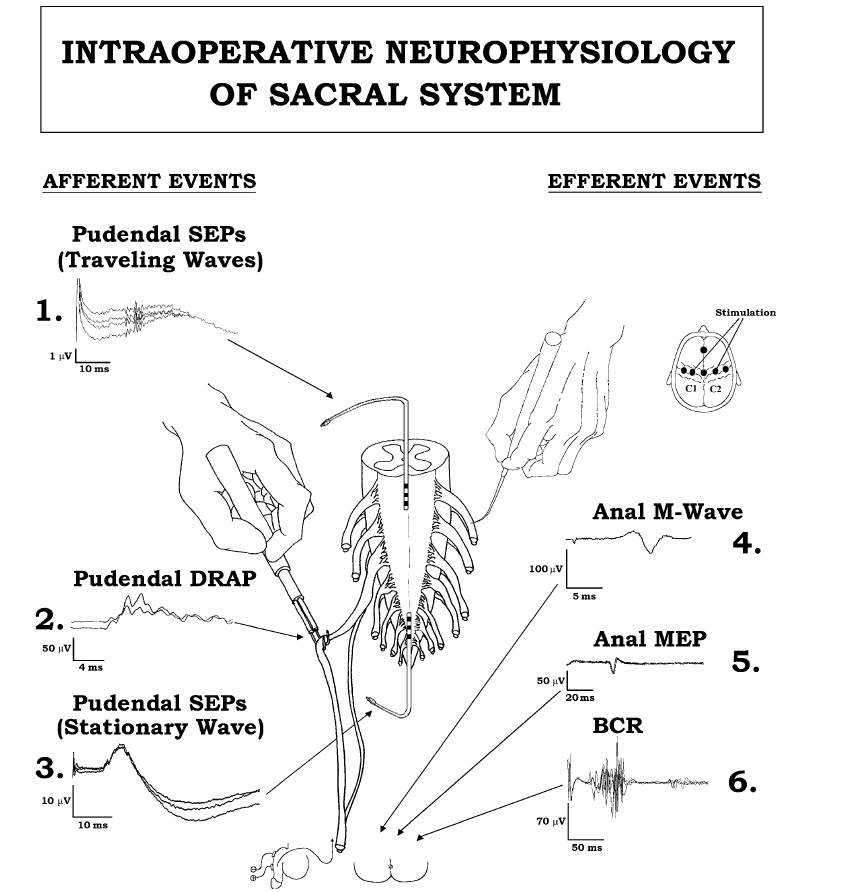 |
 |
| FIGURE 9.1 Neurophysiological events used to intraoperatively
monitor the sacral nervous
system. Left, “afferent” events after stimulation of the dorsal
penile or clitoral nerves and recording
over the spinal cord: (1) pudendal SEPs, traveling waves, (2)
pudendal DRAPs, and (3) pudendal
SEPs, stationary waves, recorded over the conus. Right,
“efferent” events: (4) anal M wave recorded
from the anal sphincter after stimulation of the S1–S3 ventral
roots, (5) anal motor-evoked potentials
recorded from the anal sphincter after transcranial electrical
stimulation of the motor cortex,
and (6) bulbocavernosus reflex obtained from the anal sphincter
muscle after electrical stimulation
of the dorsal penile or clitoral nerves. Reprinted from Deletis,
V. (2001). Neuromonitoring. In “Pediatric
neurosurgery,” 4th ed. (D. MacLeone, ed.), pp. 1204–1213. W.B.
Saunders, Philadelphia. |
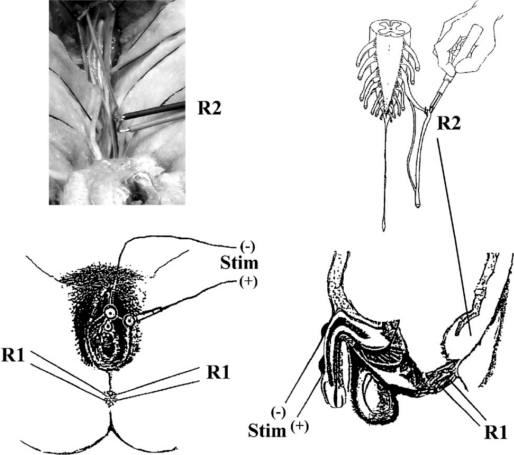
FIGURE 9.2 Lower left, position of the electrodes over the
clitoris and labia majora for the stimulation
of the dorsal clitoral nerves. Lower right, position of the
electrode for stimulating the dorsal
penile nerves. R1 = recording BCR from anal sphincter. Upper
right, schematics of recording DRAPs
with a hand-held hook electrode (R2) over the exposed dorsal
sacral roots of the cauda equina.
Upper left, intraoperative picture of DRAP recordings. |
The appropriate peripheral sensory structures that are available
for stimulating
purposes are the two adjacent dorsal penile or clitoral nerves.
These
nerves are stimulated by silver/silver chloride cup EEG-type
electrodes (EEG
electrodes) placed on the dorsal surface of the penis or
clitoris (this electrode
representing the cathode). The other electrode (anode) is placed
either distally
on the penis (1–2 cm apart from the proximal electrode) or on
the adjacent
labia (Fig. 9.2). In small children with short penises, the
anode may be attached
on the ventral side of the penis. The dorsal side of the penis
must be scrubbed
gently with Nuprep (D. O. Weaver & Co., Aurora, CO) before
placing electrodes
in order to avoid stimulus artifacts. The electrodes are filled
with electrode
cream and secured appropriately (Tegaderm; Smith and Nephew
Medical Limited,
Hull, England). The electrode sites are then bandaged with a few
layers of
gauze to prevent them from being displaced when the patient is
moved onto the
operating table. Electrode impedances should be kept below 5 kΩ.
As a general rule, stimuli of 20 mA intensity at 0.2 ms duration
have been
delivered in procedures involving stimulation of the penis or
clitoris (at various
frequencies for different measurements, up to 13.3 Hz).
Stimulation can also be performed at the level of the spinal
roots; a handheld
sterile monopolar electrode can be placed under the appropriate
roots (or
rootlets, after the root is freed from neighboring roots and
lifted outside the
spinal canal). Square wave pulses of 1 to 2 mA intensity and 0.2
ms duration
are delivered (Fig. 9.1, right).
4.2 BASIC TECHNICAL ASPECTS OF RECORDING FOR
INTRAOPERATIVE SACRAL MONITORING
Bioelectrical activity from the sacral
neuromuscular system has
up to now been recorded from the sacral dorsal spinal roots, the
spinal cord, the
somatosensory cortex, and the anal sphincter muscle.
Recordings from dorsal spinal roots (dorsal root action
potentials, DRAPs)
are obtained by hand-held sterile bipolar hook electrodes (after
the root is freed
from neighboring roots and lifted outside the spinal canal). In
this case, the
electrode closer to the point of stimulation is the G1 (active)
electrode. Epoch
lengths of 0 to 50 ms are used for these recordings (Figs. 9.1,
9.2).
Recordings of pudendal spinal somatosensory evoked responses (SSEPs,
stationary
wave) are obtained by a spinal epidural electrode placed over
the conus
(S2–S4). These potentials are generated by interneurons of the
grey matter
within the S2–S4 segments of the spinal cord. Typically 100
responses are averaged
together; epoch lengths of 0–50 ms are used (Fig. 9.1).
Recordings of pudendal spinal somatosensory evoked responses (SSEPs,
traveling
wave) are obtained by a spinal epidural electrode inserted
anywhere over
the dorsal column of the spinal cord. These potentials are
generated by pudendal
afferents traveling within the dorsal columns. Typically 100
responses are
averaged together; epoch lengths of 0–50 ms are used (Fig. 9.1).
To obtain pudendal cerebral somatosensory evoked responses (CSEPs),
Screwtype
recording electrodes are placed on the scalp 2 cm behind CZ (G1
or active
electrode) and at FZ (G2 or reference electrode), according to
the somatotopic
representation in the primary somatosensory cortex related to
the International
10–20 System of scalp electrode placement. The active electrode
is placed in the
midline because the sacral segments are represented deep within
the medial
longitudinal interhemispheric fissure. For CSEPs, 100–200 traces
are typically
averaged together. Epoch lengths of 0–200 ms are used.
To record anal sphincter (EMG) responses, either surface-type
electrodes or
hook wire electrodes can be used. Given the close anatomical
relationship
between the small sphincter muscle and neighboring larger muscle
groups,
recording must be selective (e.g., with intramuscular hook wire
electrodes) if
the stimulation technique is “nonselective” (such as in the case
of ventral root
stimulation, as a consequence of which neighboring muscles are
also excited).
When the stimulation procedure is more specific (e.g., in
bulbocavernosus or
pudendoanal reflex monitoring), the recording may be obtained
with properly
attached surface-type electrodes.
Sterile hook wire recording electrodes (Teflon-coated, 76 μm
diameter wire
with a 3 mm bare tip) are introduced into the left and right
sides of the external
anal sphincter with sterile needles; these are immediately
removed carefully
from the sphincter (the hooked wires remaining in place). The
integrity of the
electrodes can be tested by passing a short train of 50 Hz
current at 10 mA and
observing sphincter contraction (if the patient is not paralyzed
at the time of
electrode placement). The electrode impedances of these
electrodes should be
checked, although clean recordings are usually still possible
with high electrode
impedances.
The epoch length used for anal sphincter EMG recordings varies
according
to the type of response; either single or few averaged responses
can be obtained
upon stimulation (of course, the patient should not be under the
influence of a
muscle relaxant during this procedure).
4.3 SPECIFIC SACRAL NEUROMUSCULAR SYSTEM
MONITORING PROCEDURES
4.3.1 Pudendal Dorsal Root Action Potentials (DRAPs)
In the treatment of spasticity (e.g., in cerebral palsy), the
sacral roots are
increasingly being included during rhizotomy procedures.
Lang [14] demonstrated that children who underwent L2–S2
rhizotomies had
an 81% greater reduction in plantar/flexor spasticity compared
to children
who underwent only L2–S1 rhizotomies. But, as more sacral dorsal
roots have
been included in rhizotomies, neurosurgeons have experienced an
increased
rate of postoperative complications, especially with regards to
bowel and bladder
functions.
In order to spare sacral function, we attempted to identify
those sacral dorsal
roots that were carrying afferents from pudendal nerves. To do
this, we used
recordings of dorsal root action potentials (DRAPs). Patients
were anesthetized
with isoflurane, nitrous oxide, fentanyl, and a short-acting
muscle relaxant
introduced only at the time of intubation. The cauda equina was
exposed
through a T12–S2 laminotomy or laminectomy and the sacral roots
were identified
using bony anatomy. The dorsal roots were separated from the
ventral
ones, and DRAPs were recorded by a hand-held sterile bipolar
hooked electrode
(the root being lifted outside the spinal canal) (Fig. 9.2). The
DRAPs were
evoked by electrical stimulation of the penile or clitoral
nerves. One hundred
responses were averaged together and filtered between 1.5 and
2100 Hz. Each
average response was repeated to assess its reliability.
Afferent activity from the
right and left dorsal roots of S1, S2, and S3 was always
recorded, along with
occasional recordings from the S4–S5 dorsal roots. DRAP
recording was successfully
obtained in the majority of patients, the
DRAPs were present in the S2 and S3 roots bilaterally in 19
patients, whereas
in 7 patients DRAPs were also present in both S1 roots (in 8
patients they were
present in the S1 root unilaterally). However, the response in
the S1 root was
never larger than the S2 or S3 root responses. The range of
amplitudes was
2.9–18.3 μV in S1 roots, 3.2–129.9 μV in S2 roots, and 4.6–333
μV in S3 roots.
Of special relevance was the finding that in 7.6% of these
children, all afferent
activity was carried by only one S2 root (Fig. 9.3, C and F).
These findings were
confirmed by a later analysis of the results of mapping in 114
children (72 male,
42 female, mean age 3.8 years) [5]. Mapping was successful in
105 out of 114
patients; S1 roots contributed 4.0%, S2 roots 60.5%, and S3
roots 35.5% of the
overall pudendal afferent activity. The distribution of
responses was asymmetrical
in 56% of the patients (Fig. 9.3, B, C, and F). Pudendal
afferent distribution
was confined to a single level in 18% (Fig. 9.3, A), and even to
a
single root in 7.6% of patients (Fig. 9.3, C and F). Fifty-six
percent of the
pathologically responding S2 roots during rhizotomy testing were
preserved
because of the significant afferent activity, as demonstrated
during pudendal
mapping. None of the 105 patients developed long-term bowel or
bladder
complications.
All our results in the early series of dorsal root mapping with
19 patients [4]
have been confirmed by analysis of the larger series of 105
patients [5]. With
this series we showed that selective S2 rhizotomy can be
performed safely without
an associated increase in residual spasticity, while at the same
time bowel
and bladder function are preserved by performing pudendal
afferent mapping [14].
Therefore, we suggest that the mapping of pudendal afferents in
the dorsal roots
should be employed whenever these roots are considered for
rhizotomy in children
with cerebral palsy without urinary retention. Preoperative
neurourological
investigation of the children should help in making appropriate
decisions;
for example, in children with cerebral palsy with hyperreflexive
detrusor dysfunction,
sacral rhizotomy may be considered to alleviate the problem. In
any
case, intraoperative mapping of sacral afferents should make
selective surgical
approaches possible and provide the maximal benefit for children
with cerebral
palsy.
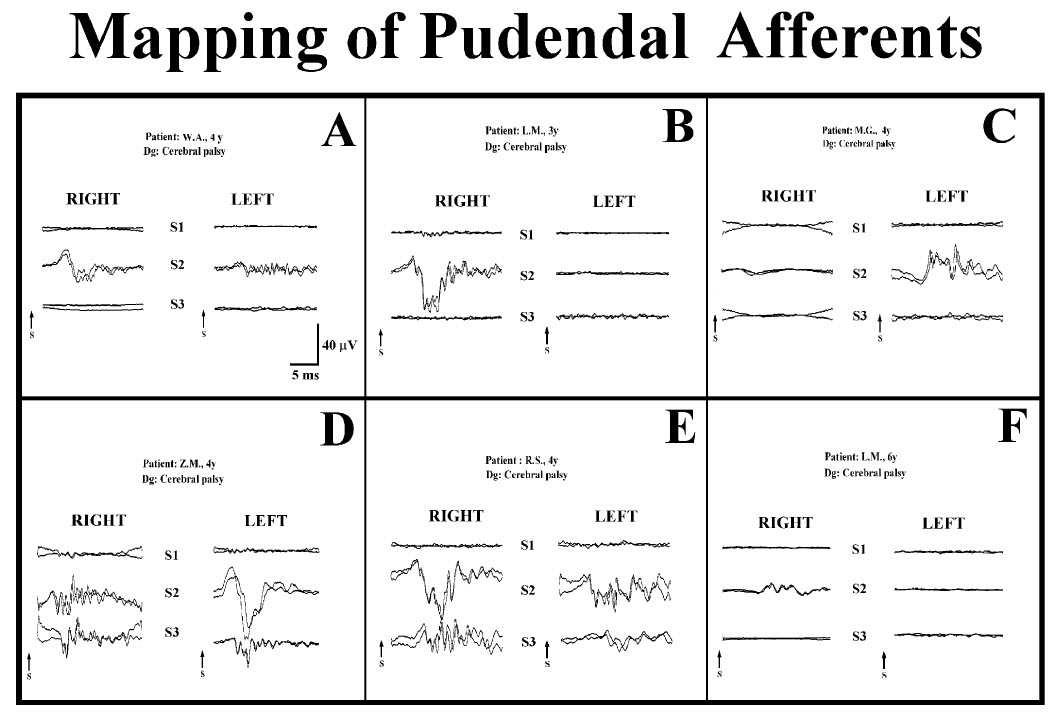 |
|
FIGURE 9.3 Six characteristic examples of DRAPs showing the
entry of a variety of pudendal nerve fibers to the spinal cord
via S1–S3 sacral roots. (A)
Symmetrical distribution of DRAPs confined to one level (S2) or
three levels (D). Asymmetrical distribution of DRAPS confined to
the right side (B), only
one root (C or F), or all roots except right S1 (E). Recordings
were obtained after electrical stimulation of bilateral
penile/clitoral nerves. |
Our results so far support the hypothesis that the root
distribution of afferent
fibers that are important for the control of micturition may be
similar to the
distribution of mucocutaneous afferent fibers from the pudendal
nerve. Only
further studies will clarify the complex functional anatomic
issues involved.
Recordings were performed in four children of both sexes, 2.5 to
7.0 years of
age. Small-amplitude (up to 1 μV) SSEPs, which were very stable,
could be
recorded with subdurally placed electrodes over the thoracic
spinal cord (traveling
waves), while a stationary wave could be recorded with a much
higher
amplitude (up to 10 μV) over the conus region. The latencies of
the spinal SEP
over the conus region ranged from 6.0 to 10.4 ms (Fig. 9.1,
left). The recordings
were made as a pilot study, and thus far only demonstrate the
ability to
obtain such recordings intraoperatively. Further employment of
this technique
showed that traveling waves are difficult to record, and
successful recording of
a stationary wave necessitates that the electrode be placed
strictly over the S2–S4
sections of the spinal cord.
4.3.3 Pudendal Cerebral Somatosensory Evoked Potentials (CSEPs)
Well-formed cerebral SEPs with amplitudes of 0.5–0.7 μV were
recorded on
dorsal penile nerve stimulation throughout spinal neurosurgical
procedures in
two adult male patients, 50 and 78 years of age. Stable P40
peaks were obtained.
The recordings were made as a pilot study and thus far only
demonstrate the
ability to obtain such recordings intraoperatively. Further
employment of this
method showed that this potential is very sensitive to
anesthetics; a formal feasibility
study has not yet been performed.
4.3.4 Anal Sphincter Motor Response Monitoring
In five children of both sexes, 2.5 to 9.0 years old, anal
sphincter muscle EMG
responses were recorded by stimulation of the ventral spinal
roots (L5, S1, S2,
S3, and S4) to identify ventral roots carrying motor fibers to
the sphincter
muscle. Recordings were obtained by surface conductive rubber
electrodes
(applied para-anally) and intramuscular hooked wire electrodes.
In surface
recordings, no unilateral responses could be identified, and
responses were
also obtained on stimulation of the L5 and S1 roots (on
stimulation of L5 and
S1 roots no adequate responses could be discerned from
simultaneous recordings
from intramuscular electrodes). The surface recorded responses
were recognized
as “nonspecific” (derived from neighboring muscles, most
probably
glutei [15]).
The latency of surface recorded responses was, as a rule,
shorter than the
latency of responses obtained from intramuscular electrodes,
which was between
5 and 8 ms. On electrical stimulation of the motor cortex, anal
sphincter
responses were recorded in a large group of anesthetized
patients without pyramidal
involvement. Because of polysynaptic connections of the
corticospinal
tract to the α-motoneuron of S2–S4, these responses are
moderately sensitive
to anesthetics (Fig. 9.1).
4.3.5 Bulbocavernosus Reflex (BCR) Monitoring
Intraoperative recordings were first performed in 15
neurosurgical patients (11
males, 4 females, 2–6 years old). Patients were without sacral
dysfunction and
were anesthetized with fenatyl and propofol or nitrous oxide
without the
influence of a muscle relaxant. Recordings from the anal
sphincter were
obtained by hooked wire electrodes and were recorded as a single
response.
Very reproducible responses could be obtained on double-pulse
stimulation,
the optimum interstimulus interval being found to be 3 ms and
the optimum
stimulation rate 2.3 Hz. Continuous periods of stimulation and
recording for
up to 10 min were repeatedly performed with very reproducible
results (Fig.
9.4). The reflex response was suppressed by the administration
of isoflurane
and nitrous oxide and was completely abolished by muscle
relaxants [16]. After
this pilot study, 119 patients were tested, 38 of which
underwent surgery without
risk and 81 of which underwent surgery with risk of damage to
sacral
structures. In all, 51 adults (19 to 64 years old, 32 male and
19 female) and
68 children (24 days to 17 years old, 30 male and 38 female)
took part in the
study. Patients were anesthetized with propofol, fenatyl, or
nitrous oxide
with a short-acting relaxant. Clinically, most patients had mild
to moderate
upper motor neuron deficits in the lower extremities, and no
patient had major
urinary problems. In all patients it was possible to record
reproducible reflex
response with the previously described method. In patients
without risk to the
sacral system, only a few minutes of the responses were recorded
to test their
feasibility, whereas in the patients at risk, continuous
monitoring was conducted.
|
 |
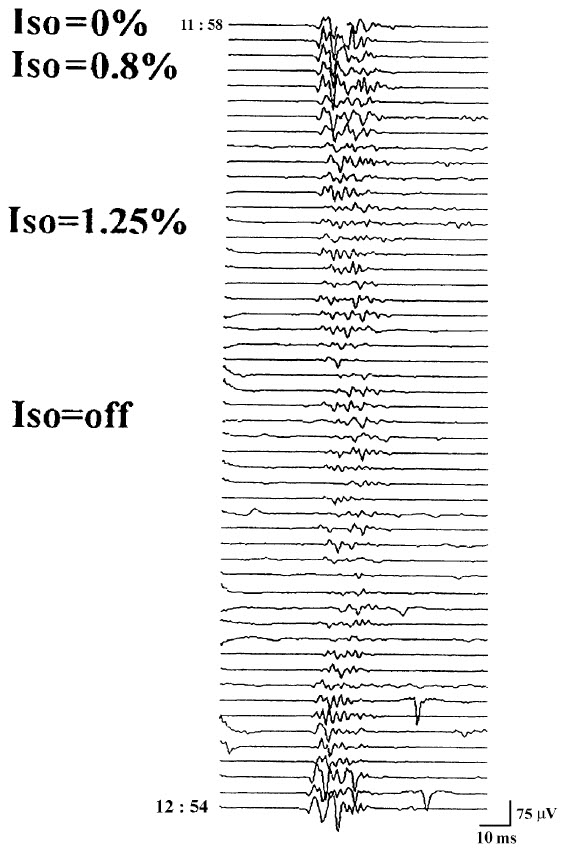 |
FIGURE 9.4 Continuous monitoring of the BCR for a period of 10
min showing the stability of
the BCR’s appearance. |
FIGURE 9.5 The influence of isoflurane (Iso) on the BCR. Note
that the response was almost completely
blocked when the concentration of 1.25% was administered for 15
min and did not recover
until almost 30 min after the isoflurane was discontinued.
Reprinted from [6]. |
The influence of volatile anesthetic was tested in 6 patients.
In 3
patients, BCR was suppressed by the administration of 1.25%
isoflurane for 15 min
(Fig. 9.5). Administration of nitrous oxide (60% inspired
concentration) for
15 min reduced the BCR in 3 patients, and the response was
completely abolished
by muscle relaxant in 2 patients [6].
Preliminary results conducted in 50 patients with a lumbosacral
tethered
cord showed no clear correlation between BCR and postoperative
sphincter control. The authors concluded that the complexity of
sphincter
innervation and segmental or suprasegmental control probably
accounts for this
discrepancy [17].
Up to now, close to 250 patients have been monitored using this
method. In
the last 100 patients, a train of four consecutive stimuli [7]
was used rather than
a double stimulus, providing even more robust responses. Some
problems have
been encountered in female patients in whom the stimulation
method is not as
robust as required; this problem needs particular attention from
the technician.
5 DISCUSSION AND CONCLUSIONS
The impetus to include intraoperative monitoring of sacral
nervous structures
came from concerns over optimizing care for children with
cerebral palsy.
These were successfully treated for their spasticity by
performing selective
dorsal rhizotomy; surgeons have always sought to minimize the
side-effects of
this procedure while maintaining its benefits (benefits being a
reduction in
muscle hypertonia, the side-effects being a partial or complete
loss of sensory
modalities). The first sacral dorsal roots have always been a
target for rhizotomy
in this procedure, but the S2 dorsal roots have become
increasingly considered
for rhizotomy. Not to treat the S2 dorsal roots is to leave
potentially abnormal
reflexive circuits that will continue to drive spasticity in the
musculature of the
leg. For this reason, the lesion zone was extended to include S2
dorsal roots,
and the result was a greater reduction in spasticity as compared
with children
in whom the lesion was extended only to the S1 segment.
Unfortunately, the
extension to include S2 roots was also associated with disorders
in micturition.
In one group of patients, before DRAP mapping was introduced,
24% experienced
urinary retention (which was only transient in most children).
Although
most of these children had both the left and right S2 roots cut,
2 of the children
had only one S2 root cut, and they also experienced retention.
The selective
dorsal rhizotomy procedure should be performed in young children
who are
certainly too young to assess sexual function; they are even too
young to be
completely confident that all symptomatic complaints regarding
micturition or
defecation and perineal sensation were being relayed. It was
this concern over
the preservation of genitourinary afferent function that led us
to develop the
technique of intraoperative neurophysiological identification of
the sacral roots
responsible for perineal sensation. In the first 31 children,
neurophysiological
identification of roots and rootlets carrying afferent activity
from the penile or
clitoral nerves led to zero micturition disturbances and allowed
for rhizotomy
of S2 roots or rootlets not carrying such afferent activity.
Therefore, maximum
possible antispastic effects could be achieved. The particularly
important lesson
we learned by doing very systematic recordings in S1–S2 and S3
roots bilaterally
(and in some children also S4 and S5 roots) is that although
most of our
patients showed evidence of pudendal afferents in S2 and S3
roots bilaterally,
about half of them also showed evidence of some afferent
activity in S1 (either
unilaterally or bilaterally). In slightly more than half of our
patients, the root
potentials were symmetrical, but the pudendal afferents of many
were irregularly
distributed across the sacral roots. In a few of the children
the afferentactivity was confined to a single root (either S2 or S3 root).
Since this may be
the most important afferent contribution from the genitourinary
area, even if
only the S2 dorsal roots are checked for relevant afferent
activity, they should
not be sacrificed if they show any such activity. The finding of
asymmetrical distribution
of fibers that may be confined to a single root is consistent
with the
work of Junemann et al. [18], who found that the majority of
motor fibers for
the urethral sphincter are carried by a single variable lower
sacral root. Therefore,
for some patients undergoing an operation in the area of the
cauda equina,
the sacrifice of one single root may have dire functional
consequences.
For other neurosurgical procedures in the lumbosacral spinal
canal (e.g.,
the release of a tethered cord or the removal of a tumor), other
authors have
proposed, and in a small series have performed, the
identification of motor and
sensory nerve roots. This has been achieved through continuous
monitoring of
electromyographic activity in muscles innervated by lumbosacral
segments, and
through monitoring of tibial nerve somatosensory evoked
potentials [19]. In
addition, monitoring pudendal SEPs should provide very relevant
complementary
information, and, as we have demonstrated, should not be
technically
demanding if the structures are preserved preoperatively.
Kothbauer et al. [19]
claim that intraoperative recordings saved operating time by
allowing the surgeon
more rapid and decisive preparation than would be possible on an
anatomical basis alone, and they also gave the impression that
the procedures
were safer. The recordings of spontaneous anal sphincter
electromyographic
activity during such operations have been described earlier
[20]. To accomplish
the two goals of neurophysiology (i.e., first, immediate
identification of structures
as functional nervous tissue and their distinction from other
tissue; and
second, continuous monitoring of the function of the relevant
nervous structures),
a battery of methods needs to be applied, and a whole set of
structures
needs to be assessed bilaterally. Therefore, both the lower
sacral segments and
sphincter muscles, and also the upper sacral segments and the
lumbar segments,
need to be included. Also, all these segments and several
functional
modalities need to be monitored more or less simultaneously. The
appropriate
setup for each surgical situation would need to be selected on
the basis of
anatomical and physiological considerations, and a compromise
between the
possible and the necessary would be sought.
Other authors have claimed that continuous EMG recording of
bursts or
trains of motor unit potentials or repetitive neurotonic
discharges elicited by
injury to the peripheral motor fibers have correlated with
postoperative transient
or permanent neurological deficit [21, 22] in the area of facial
nerves. The predictive
value of these “manipulation-evoked” discharges is only based on
empirical
data, but has been proposed to also be of value in the
lumbosacral segments
[19]. The electrophysiological identification of motor nerves is
already an integral
part of cranial nerve surgery [22] and should also provide a
similar service
in the region of the cauda equina.
Up to now, only recordings of DRAPs have been made in a large
number of
subjects (to identify sacral roots carrying genitourinary
afferents), and the electrophysiological
procedure decreased postoperative voiding disturbances [4, 5].
We propose, however, that the other intraoperative
electrophysiological recordings
of the sacral neuromuscular system that have been described
(spinal SEP,
CMAP of sphincter muscles, bulbocavernosus reflex recordings)
should prove
relevant in surgeries involving the sacral roots, the cauda
equina, and the conus
and should aid the surgeon in preventing inadvertent damage to
these structures.
These other procedures have not, however, been performed in an
adequate
number of patients, and their relevance cannot yet be appraised.
The
most interesting question is whether monitoring of the
bulbocavernosus reflex
for conus and cauda equina surgery could, as we are proposing,
replace and
even improve upon separate monitoring of motor and sensory
fibers of the
lower sacral roots.
As for surgeries involving the spinal cord above the conus, the
procedures
of pudendal SSEPs, cortical SEPs, and (possibly) motor evoked
potentials of the
anal sphincter muscle might be interesting in some selected
patient groups.
These would include groups in whom the preservation of sacral
function may
be particularly important (for instance, in scoliosis surgery in
patients with
advanced neuromuscular diseases who have heavily compromised
motor function
but no sacral dysfunction).
In other contexts, authors have argued that recordings from the
anal sphincter
cannot be taken as completely adequate information on the
functional status
of the urethral sphincter [18]. Since innervation of both
sphincters originates
from the same sacral segments (which also provide innervation
for the detrusor),
the monitoring of sphincter ani should generally mirror the
function of
relevant urethral structures.
As previously stated, the relatively limited experience with the
monitoring
of sacral structures cannot as yet prove that surgeries
accompanied by such
monitoring are easier and safer. For the time being, there is
anecdotal evidence
that difficult surgical decisions that have relied on the
results of intraoperative
neurophysiological measurements have not resulted in unexpected
neurological
deficits. Although the techniques described clearly provide the
surgeon with
additional information about nerve location and function, the
value of these
techniques must be further defined and documented. As Daube [23]
suggests,
it will be necessary to demonstrate that these techniques indeed
save
operative time, save anesthesia time, or—most
importantly—improve outcomes.
It will be difficult to demonstrate this without doing a
randomized study
of patients who are undergoing surgery with and without such
monitoring
techniques.
|
 |
|