![]() Magnetic
resonance imaging
(MRI), nuclear magnetic resonance
imaging (NMRI), or magnetic
resonance tomography (MRT)
Magnetic
resonance imaging
(MRI), nuclear magnetic resonance
imaging (NMRI), or magnetic
resonance tomography (MRT)
is a medical imaging
technique used in radiology to visualize
detailed internal structures. MRI makes use of
the property of nuclear magnetic resonance (NMR)
to image nuclei of atoms inside the body. An MRI
machine uses a powerful magnetic field to align
the magnetization of some atoms in the body, and
radio frequency fields to systematically alter
the alignment of this magnetization. This causes
the nuclei to produce a rotating magnetic field
detectable by the scanner—and this information
is recorded to construct an image of the scanned
area of the body. Strong magnetic field
gradients cause nuclei at different locations to
rotate at different speeds. 3-D spatial
information can be obtained by providing
gradients in each direction.
MRI provides good contrast between the different
soft tissues of the body, which makes it
especially useful in imaging the brain, muscles,
the heart, and cancers compared with other
medical imaging techniques such as computed
tomography (CT) or X-rays. Unlike CT scans or
traditional X-rays, MRI uses no ionizing
radiation.
|
|
History |
|
In the 1950s, Herman Carr
reported on the creation of a one-dimensional MR
image. Paul Lauterbur expanded on Carr's
technique and developed a way to generate the
first MRI images, in 2D and 3D, using gradients.
In 1973, Lauterbur published the first nuclear
magnetic resonance image, and the first
cross-sectional image of a living mouse was
published in January 1974. Nuclear magnetic
resonance imaging is a relatively new technology
first developed at the University of Nottingham,
England. Peter Mansfield, a physicist and
professor at the university, then developed a
mathematical technique that would allow scans to
take seconds rather than hours and produce
clearer images than Lauterbur had.
In a 1971 paper in the journal Science, Dr.
Raymond Damadian, an Armenian-American
physician, scientist, and professor at the
Downstate Medical Center State University of New
York (SUNY), reported that tumors and normal
tissue can be distinguished in vivo by nuclear
magnetic resonance ("NMR"). He suggested that
these differences could be used to diagnose
cancer, though later research would find that
these differences, while real, are too variable
for diagnostic purposes. Damadian's initial
methods were flawed for practical use, relying
on a point-by-point scan of the entire body and
using relaxation rates, which turned out to not
be an effective indicator of cancerous tissue.
While researching the analytical properties of
magnetic resonance, Damadian created the world's
first magnetic resonance imaging machine in
1972. He filed the first patent for an MRI
machine, U.S. patent #3,789,832 on March 17,
1972, which was later issued to him on February
5, 1974. As the National Science Foundation
notes, "The patent included the idea of using
NMR to 'scan' the human body to locate cancerous
tissue. However, it did not describe a method
for generating pictures from such a scan or
precisely how such a scan might be done.
Damadian along with Larry Minkoff and Michael
Goldsmith, subsequently went on to perform the
first MRI body scan of a human being on July 3,
1977. These studies performed on humans were
published in 1977.
In recording the history of MRI, Mattson and
Simon (1996) credit Damadian with describing the
concept of whole-body NMR scanning, as well as
discovering the NMR tissue relaxation
differences that made this feasible.
Reflecting the fundamental importance and
applicability of MRI in medicine, Paul Lauterbur
of the University of Illinois at
Urbana-Champaign and Sir Peter Mansfield of the
University of Nottingham were awarded the 2003
Nobel Prize in Physiology or Medicine for their
"discoveries concerning magnetic resonance
imaging". The Nobel citation acknowledged
Lauterbur's insight of using magnetic field
gradients to determine spatial localization, a
discovery that allowed rapid acquisition of 2D
images. Mansfield was credited with introducing
the mathematical formalism and developing
techniques for efficient gradient utilization
and fast imaging. The actual research that won
the prize was done almost 30 years before, while
Paul Lauterbur was at Stony Brook University in
New York.
The award was vigorously protested by Raymond
Vahan Damadian, founder of FONAR Corporation,
who claimed that he invented the MRI, and that
Lauterbur and Mansfield had merely refined the
technology. An ad hoc group, called "The Friends
of Raymond Damadian", took out full-page
advertisements in the New York Times and The
Washington Post entitled "The Shameful Wrong
That Must Be Righted", demanding that he be
awarded at least a share of the Nobel Prize.
Also, even earlier, in the Soviet Union,
Vladislav Ivanov filed (in 1960) a document with
the USSR State Committee for Inventions and
Discovery at Leningrad for a Magnetic Resonance
Imaging device, although this was not approved
until the 1970s. In a letter to Physics Today,
Herman Carr pointed out his own even earlier use
of field gradients for one-dimensional MR
imaging.
|
|
MRI Scanner Anatomy |
|
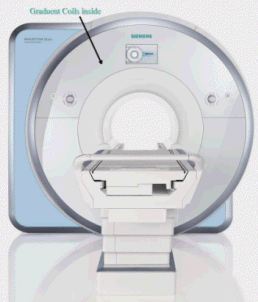
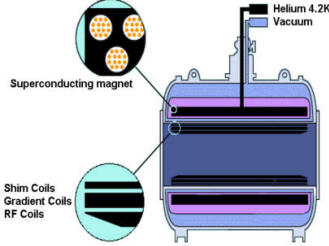
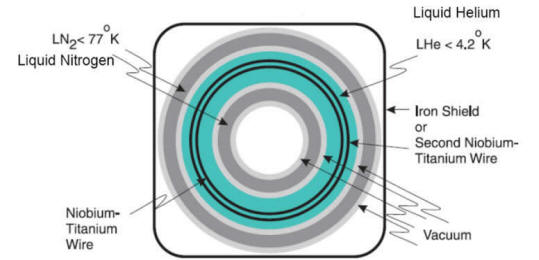
| A helium-cooled superconducting
magnet generates the static field. Always on: only quench field in emergency. niobium titanium wire. Coils allow us to : Make static field homogenous (shims: solenoid coils) Briefly adjust magnetic field (gradients) Transmit, record RF signal (RF coils: antennas) |
 |
 |
 |
 |
|
|
|
How MRI works |
|
The body is largely composed
of water molecules. Each water molecule has two
hydrogen nuclei or protons. When a person is
inside the powerful magnetic field of the
scanner, the magnetic moments of some of these
molecules become aligned with the direction of
the field. A radio frequency transmitter is
briefly turned on, producing a further varying
electromagnetic field. The photons of this field
have just the right energy, known as the
resonance frequency, to be absorbed and flip the
spin of the aligned protons in the body. The
frequency at which the protons resonate depends
on the strength of the applied magnetic field.
After the field is turned off, those protons
which absorbed energy revert to the original
lower-energy spin-down state. Now a hydrogen
dipole has two spins, 1 high spin and 1 low. In
low spin both dipole and field are in parallel
direction and in high spin case it is
antiparallel. They release the difference in
energy as a photon, and the released photons are
detected by the scanner as an electromagnetic
signal, similar to radio waves.
As a result of conservation of energy, the
resonant frequency also dictates the frequency
of the released photons. The photons released
when the field is removed have an energy — and
therefore a frequency — which depends on the
energy absorbed while the field was active. It
is this relationship between field-strength and
frequency that allows the use of nuclear
magnetic resonance for imaging. An image can be
constructed because the protons in different
tissues return to their equilibrium state at
different rates, which is a difference that can
be detected. Five different tissue variables —
spin density, T1 and T2 relaxation times and
flow and spectral shifts can be used to
construct images. By changing the settings on
the scanner, this effect is used to create
contrast between different types of body tissue
or between other properties, as in fMRI and
diffusion MRI.
The 3D position from which photons were released
is learned by applying additional fields during
the scan. This is done by passing electric
currents through specially-wound solenoids,
known as gradient coils. These fields make the
magnetic field strength vary depending on the
position within the patient, which in turn makes
the frequency of released photons dependent on
their original position in a predictable manner,
and the original locations can be mathematically
recovered from the resulting signal by the use
of inverse Fourier transform.
Contrast agents may be injected intravenously to
enhance the appearance of blood vessels, tumors
or inflammation. Contrast agents may also be
directly injected into a joint in the case of
arthrograms, MRI images of joints. Unlike CT,
MRI uses no ionizing radiation and is generally
a very safe procedure. Nonetheless the strong
magnetic fields and radio pulses can affect
metal implants, including cochlear implants and
cardiac pacemakers. In the case of cochlear
implants, the US FDA has approved some implants
for MRI compatibility. In the case of cardiac
pacemakers, the results can sometimes be lethal,
so patients with such implants are generally not
eligible for MRI.
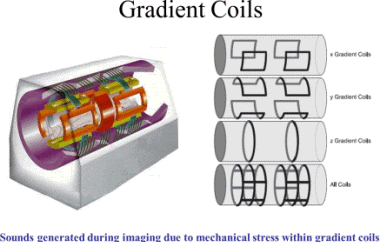
Since the gradient coils are within the bore of
the scanner, there are large forces between them
and the main field coils, producing most of the
noise that is heard during operation. Without
efforts to damp this noise, it can approach 130
decibels (dB) with strong fields.
MRI is used to image every part of the body, and
is particularly useful for tissues with many
hydrogen nuclei and little density contrast,
such as the brain, muscle, connective tissue and
most tumors.
|
|
3 tesla MRI vs 1.5 tesla. |
|
High field strength machines provide faster and
better scanning. As good as 1.5–tesla MR images
are, they just can’t characterize some subtle
abnormalities as well as 3 tesla can. Take the
1.5–tesla scan of a 14–year–old seizure victim
that was normal except for an area of
ventricular dilation. It wasn’t until
radiologists at Massachusetts General Hospital
performed 3–tesla MRI that they could spot
heterotopic gray matter associated with a
developmental problem.
High field strength MRI delineates structures
that have not been seen before, such as blood
vessels as small as 200 to 300 microns, drilling
down to the ultra–structural level. But 3–tesla
MRI is more than a glorified, high–tech
microscope. According to radiologists who have
been testing high field strength MRI in clinical
settings, 3–tesla machines can do anything a
workhorse 1.5–tesla scanner can, and do it
faster and better. Conventional brain imaging at
3 tesla can be completed in the same exam time
and can achieve a higher signal–to–noise ratio
than at 1.5 tesla.
With a three– to four–fold higher SNR, 3–tesla
MRI more precisely localizes areas of
activation, enabling accurate mapping of brain
function in patients more than 90% of the time.
Three–tesla MRI also makes potentially
insensitive techniques clinically robust.
In the body, the SNR of a
3–tesla body coil is about the same as that of a
1.5–tesla phased–array coil. Adding a surface
coil gives much more signal–to–noise headroom,
which allows faster image acquisition and
patient throughput or higher image resolution,
revealing fine anatomic details and physiologic
parameters.
But while 3–tesla clinical systems have the same
user interface as their 1.5–tesla predecessors,
they take a little getting used to. Radiologists
need to adjust to 3 tesla’s longer T1–weighted
imaging, modify doses of contrast medium, and
take care not to misinterpret data because of
differences in contrast. So there is a learning
curve, although not a steep one. Current opinion
is that 3–tesla MRI will become the clinical
standard, initially in neuroimaging, and
eventually throughout the body.
![]() 3–Tesla in the
Brain
3–Tesla in the
Brain
The demand for high–performance imaging is
demonstrating clear medical benefit in cases
where we can’t see lesions well enough with 1.5
tesla, such as scanning for developmental
abnormalities or performing high–resolution MR
angiography, diffusion or perfusion imaging. The
biggest payoff for 3 tesla is in presurgical
planning to avoid invasive angiography or direct
cortical mapping at the time of surgery. Coupled
with anatomic detail, 3–tesla perfusion studies
plot a patient’s physiology up to the edge of a
lesion, and diffusion tensor images trace
white–matter tracks that must be preserved.
The signal changes that we look at with blood
oxygenation level–dependent contrast at 1.5
tesla is on the order of 1% to 2%. That signal
change goes to 3% to 5% at 3 tesla, meaning that
you can do reliable individual patient mapping
and interpretation.
A second major clinical neuroimaging category
for 3–tesla MRI involves monitoring the effects
of rehabilitation in patients with cognitive
impairment after successful acute treatment of
acquired brain injury. Some patients do not
return to normal after acquired brain injury
because of neurological damage to cognitive
functions. A patient can’t read, for example,
because a traumatic head injury has interfered
with the visual space processing that allows
coordination of eye movements.
It’s quite clear that patients with cognitive
difficulties after acute brain injury have
abnormal patterns of brain activation. As they
go through cognitive behavioral therapy, their
patterns of activation return toward normal, and
we can monitor how treatment engages cognitive
processes to improve performance.
Nevertheless, some had been unable to run a full
3–tesla neuroradiology schedule with a scanner
limited to the head. Now that scanner has been
equipped with a neurovascular receive–only coil,
one can extend high field strength imaging to
the neck and examine the blood vessels feeding
the brain as well as the circle of Willis. When
a phased array spine coil is designed, he will
have a full–service, 3–tesla neuroradiology
operation that includes the thoracic and lumbar
spine.
![]() Whole–Body 3
Tesla
Whole–Body 3
Tesla
With installation of the
first clinical whole–body, 3–tesla MRI, it has
been used like 1.5–tesla machines, but even
beyond. Because of the enhanced resolution of
images and the ability to factor in physiology,
3–tesla imaging has not only captured finer
details of anatomy, it has improved the
separation of spectroscopic peaks of chemical
species in the prostate. It has also identified
small lesions in the breast, evaluated
peripheral vascular disease, and tackled
phosphorus imaging. Extremely rapid imaging at 3
tesla will eliminate the need for breath–holding
and open the door to routine screening in the
abdomen where safety, speed, and throughput are
paramount.
The abdomen with noncontrast 3–tesla MRI will
exceed the capacity of noncontrast CT to detect
early disease, and without exposing patients to
ionizing radiation. The machine also is easy to
use because you don’t have to position external
coils. The patient just gets on the table in the
scanner, you do your exam, and then take them
out.
High field strength MRI can perform super–fast
imaging without sacrificing image quality. Speed
in the past was achieved by gradient structure,
but now other technologies such as parallel
imaging remove hardware constraints.
Parallel imaging, used in sensitivity encoding
(SENSE), can speed up any MRI sequence by a
factor of two, three, four, or more, depending
on the number of elements in the RF coil.
The SENSE factor on standard 1.5–tesla machines
can double sequencing speed at most. Experience
with SENSE and 3–tesla MRI indicates that
parallel imaging can raise speeds by a factor of
six or eight, bringing metabolic mapping within
reach.
Spectroscopic imaging usually takes 10 to 12
minutes. With SENSE techniques and 3–tesla MRI,
you increase resolution and still have a decent
exam time. A minute or two, and your spectra are
done.
![]() Mechanical Machinations
Mechanical Machinations
The first round of 3–tesla MR
scanners, designed for research, didn’t have
clinical front ends, and that was just fine for
investigators who love to tinker with innovative
imaging protocols. But for some clinicians,
3–tesla prototypes reduced productivity because
they lacked access in the past to turbo
spin–echo, fast spin–echo, and other sequences.
New 3–tesla machines from most manufacturers,
however, have standardized software platforms
across an entire MR product line. So most
3–tesla machines have the same user interfaces
and parameters as 1.5–tesla scanners.
T2–weighted imaging is exactly the same at 1.5
tesla and 3 tesla; MR angiography is pretty much
the same at both field strengths; and other
pulse sequences have been redesigned to operate
efficiently at 3 tesla. Radiologists therefore
can often take an imaging protocol, recompile
the pulse sequences on the 3–tesla unit, and
take advantage of high field strength.
The transition from 1.5– to 3–tesla body imaging
has actually been easier from an engineering as
well as an optimization perspective because
manufacturers have overcome the problem of the
specific absorption rate (SAR).
The concern with 3–tesla MRI was that with
increased radio–frequency exposure, you would
ultimately run into physiologic human
limitations that would restrict the number of
slices or the speed at which you could scan. But
with software and body–coil engineering designs,
clinical 3–tesla MRIs can match virtually all
1.5–tesla SAR performance standards.
Manufacturers have optimized entire 3–tesla
systems–not just field strength–to preserve the
benefits of extra signal to noise. 3–tesla
scanner had such lackluster gradient performance
that could get better diffusion images at 1.5
tesla. But new 3–tesla machines have much higher
slew rates of 400 millitesla per msec and higher
gradient performance of at least 4 gauss per cm.
High field strength MRI nevertheless is not a
simple plug–in. Radiologists can’t take every
single 1.5–tesla protocol, install it into a
3–tesla machine, and get reliable, consistent
protocols.
Protocols have to be modified because of the
higher RF. RF power more or less goes with the
square of the field strength. That means we use
four times more RF power, which makes a
difference with many sequences because of
specific absorption rate limitations.
T1–weighted imaging requires special attention
because it is slightly longer at 3 tesla and
therefore produces greater inflow enhancement
and background suppression. Radiologists
consequently need to modify TR and TE and alter
the dose of contrast medium. The amount of
contrast medium can be reduced by half.
Full–contrast doses are disturbingly black and
white, so you have to adjust the contrast if you
are doing hard–copy films, or to window through
the dynamic range to get the full benefit on
PACS.
Because of the difference in contrast between
1.5 and 3 tesla, radiologists also need to
refine their interpretation of data. Contrast
enhancement of the dura, which indicates
meningitis or carcinomatosis at 1.5 tesla, is
common on 3–tesla images. If radiologists are
not careful, they can mistake normal tissue for
white–matter disease.
The increased SNR of high field strength MRI
offers radiologists the luxury of choosing
between maximizing throughput or image
resolution.
Extra SNR is a little like currency.
Radiologists can choose to spend it on
extra–high–resolution scans; they can spend it
on shorter exam times by not bumping up the
matrix size; or they can spend it on different
contrast–to–noise ratios that they couldn’t do
before.
Routine brain imaging at 3 tesla can be done
more efficiently than at 1.5 tesla. A standard
3–tesla MRI brain scan, including fast
spin–echo, FLAIR, gradient–echo, and
high–resolution pre– and postcontrast whole
brain imaging, can be completed in 30 minutes
and still leave time to turn the table around.
![]() What’s Next?
What’s Next?
Over the next few years,
3–tesla MRI likely will follow the same
trajectory as any new technology. A group of
leaders will always be ready to take on new
technology. In this case, it’s the major medical
schools and university–based hospitals. In a few
years, the rest will follow.
Enough data today support 3–tesla MRI as the
principal technology for brain imaging in
academic medical centers that balance clinical
applications and research. And high field
strength MRI, at least in the short term, will
have targeted applications in the body to
provide highly detailed images and spectroscopy
of small organs, such as the prostate, or
minuscule lesions that lie buried behind complex
anatomy.
Applications
In clinical practice, MRI is used to distinguish pathologic tissue (such as a brain tumor) from normal tissue. One advantage of an MRI scan is that it is harmless to the patient. It uses strong magnetic fields and non-ionizing radiation in the radio frequency range, unlike CT scans and traditional X-rays, which both use ionizing radiation.
While CT provides good spatial resolution (the ability to distinguish two separate structures an arbitrarily small distance from each other), MRI provides comparable resolution with far better contrast resolution (the ability to distinguish the differences between two arbitrarily similar but not identical tissues). The basis of this ability is the complex library of pulse sequences that the modern medical MRI scanner includes, each of which is optimized to provide image contrast based on the chemical sensitivity of MRI.
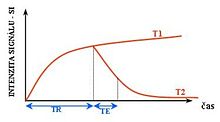
For example, with particular values of the echo time (TE) and the repetition time (TR), which are basic parameters of image acquisition, a sequence takes on the property of T2-weighting. On a T2-weighted scan, water- and fluid-containing tissues are bright (most modern T2 sequences are actually fast T2 sequences) and fat-containing tissues are dark. The reverse is true for T1-weighted images. Damaged tissue tends to develop edema, which makes a T2-weighted sequence sensitive for pathology, and generally able to distinguish pathologic tissue from normal tissue. With the addition of an additional radio frequency pulse and additional manipulation of the magnetic gradients, a T2-weighted sequence can be converted to a FLAIR sequence, in which free water is now dark, but edematous tissues remain bright. This sequence in particular is currently the most sensitive way to evaluate the brain for demyelinating diseases, such as multiple sclerosis.
The typical MRI examination consists of 5–20 sequences, each of which are chosen to provide a particular type of information about the subject tissues. This information is then synthesized by the interpreting physician.
Basic MRI scans
T1-weighted MRI
T1-weighted scans are a standard basic scan, in particular differentiating fat from water - with water darker and fat brighter use a gradient echo (GRE) sequence, with short TE and short TR. This is one of the basic types of MR contrast and is a commonly run clinical scan. The T1 weighting can be increased (improving contrast) with the use of an inversion pulse as in an MP-RAGE sequence. Due to the short repetition time (TR) this scan can be run very fast allowing the collection of high resolution 3D datasets. A T1 reducing gadolinium contrast agent is also commonly used, with a T1 scan being collected before and after administration of contrast agent to compare the difference. In the brain T1-weighted scans provide good gray matter/white matter contrast; in other words, T1-weighted images highlight fat deposition.
T2-weighted MRI
T2-weighted scans are another basic type. Like the T1-weighted scan, fat is differentiated from water - but in this case fat shows darker, and water lighter. For example, in the case of cerebral and spinal study, the CSF (cerebrospinal fluid) will be lighter in T2-weighted images. These scans are therefore particularly well suited to imaging edema, with long TE and long TR. Because the spin echo sequence is less susceptible to inhomogeneities in the magnetic field, these images have long been a clinical workhorse.
T*2-weighted MRI
T*2 (pronounced "T 2 star") weighted scans
use a gradient echo (GRE) sequence, with long
TE and long TR.
The gradient echo sequence used does not have
the extra refocusing pulse used in
spin echo so it is subject to additional
losses above the normal T2
decay (referred to as T2′),
these taken together are called T*
2. This also makes it more prone to
susceptibility losses at air/tissue boundaries,
but can increase contrast for certain types of
tissue, such as venous blood.
Spin density weighted MRI
Spin density, also called proton density, weighted scans try to have no contrast from either T2 or T1 decay, the only signal change coming from differences in the amount of available spins (hydrogen nuclei in water). It uses a spin echo or sometimes a gradient echo sequence, with short TE and long TR.
|
|
Acquisition Parameters |
|
| Acquisition Parameters1) | ||
| 2D | Number of slices | 1 − 128 (steps of 1) |
| Slice order | Sequential or interleaved | |
| 3D Slabs / Partitions | Number of 3D partitions for matrix 256 × 256 | 4 − 512 |
| Number of 3D Slabs (3D volumes) | 1 − 128 (steps of 1) | |
| Acquisition Matrix | Frequency encoding (true imaging matrix without interpolation or oversampling) | 64 − 1024 (in steps of 2; sequence dependent) |
| Phase encoding | 32 − 1024 (in steps of 1) | |
| Reduced Matrix | Phase resolution (rectangular matrix) | 32 × n … n × n (steps of 1) |
| Slice resolution (3D volumes) | 50–100 % | |
| Partial Fourier Imaging | Phase partial Fourier (Half Fourier) | 4 / 8 − 1 (steps of 1 / 8) |
| Read partial Fourier (asymmetric echo) | Selectable | |
| Slice partial Fourier (3D volumes) | 5 / 8 − 1 (steps of 1 / 8) | |
| Rectangular Field of View | In phase encoding direction | 3 − 100 % |
| Averaging | Number of data acquisitions | 1 − 32 (steps of 1) |
| Averaging mode | Short term, Long term (LOTA) | |
| Oversampling | Read oversampling | 100% standard |
| Phase oversampling | 0 − 100 % (steps of 12.5 %) | |
| Slice oversampling (3D volumes) | 0 − 100 % (steps of 12.5 %) | |
| Interpolation | In plane interpolation | Selectable (factor of 2) |
| 3D interpolation (3D volumes) | Selectable (up to factor of 2) | |
| Serial Acquisitions | Number of repeated scans | With constant delay times 1 − 4096 |
| With different delay times 1 − 65 | ||
| Swap | Exchange of read-out and phase-encoding direction | Yes |
| Slice orientation | Slice orientation for 2D and 3D scans | Transverse, sagittal, coronal, oblique, double oblique (steps of 0.1°) |
| Multi-slice multi-angle (simultaneously) | Yes | |
1)
Combinations of the parameters stated are not always possible; some parameters may depend on optional application packages
Specialized MRI scans
Diffusion MRI
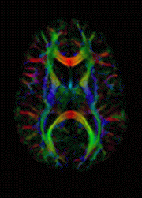
Diffusion MRI measures the diffusion of water molecules in biological tissues.[25] In an isotropic medium (inside a glass of water for example), water molecules naturally move randomly according to turbulence and Brownian motion. In biological tissues however, where the Reynolds number is low enough for flows to be laminar, the diffusion may be anisotropic. For example, a molecule inside the axon of a neuron has a low probability of crossing the myelin membrane. Therefore the molecule moves principally along the axis of the neural fiber. If it is known that molecules in a particular voxel diffuse principally in one direction, the assumption can be made that the majority of the fibers in this area are going parallel to that direction.
The recent development of diffusion tensor imaging (DTI)[7] enables diffusion to be measured in multiple directions and the fractional anisotropy in each direction to be calculated for each voxel. This enables researchers to make brain maps of fiber directions to examine the connectivity of different regions in the brain (using tractography) or to examine areas of neural degeneration and demyelination in diseases like Multiple Sclerosis.
Another application of diffusion MRI is diffusion-weighted imaging (DWI). Following an ischemic stroke, DWI is highly sensitive to the changes occurring in the lesion.[26] It is speculated that increases in restriction (barriers) to water diffusion, as a result of cytotoxic edema (cellular swelling), is responsible for the increase in signal on a DWI scan. The DWI enhancement appears within 5–10 minutes of the onset of stroke symptoms (as compared with computed tomography, which often does not detect changes of acute infarct for up to 4–6 hours) and remains for up to two weeks. Coupled with imaging of cerebral perfusion, researchers can highlight regions of "perfusion/diffusion mismatch" that may indicate regions capable of salvage by reperfusion therapy.
Like many other specialized applications, this technique is usually coupled with a fast image acquisition sequence, such as echo planar imaging sequence.
Magnetization Transfer MRI
Magnetization transfer (MT) refers to the transfer of longitudinal magnetization from free water protons to hydration water protons in NMR and MRI.
In magnetic resonance imaging of molecular solutions, such as protein solutions, two types of water molecules, free (bulk) and hydration (bound), are found. Free water protons have faster average rotational frequency and hence less fixed water molecules that may cause local field inhomogeneity. Because of this uniformity, most free water protons have resonance frequency lying narrowly around the normal proton resonance frequency of 63 MHz (at 1.5 teslas). This also results in slower transverse magnetization dephasing and hence longer T2. Conversely, hydration water molecules are slowed down by interaction with solute molecules and hence create field inhomogeneities that lead to wider resonance frequency spectrum.
In free liquids, protons, which may be viewed classically as small magnetic dipoles, exhibit translational and rotational motions. These moving dipoles disturb the surrounding magnetic field however on long enough time-scales (which may be nanoseconds) the average field caused by the motion of protons is zero. This is known as “motional averaging” or narrowing and is characteristic of protons moving freely in liquid. On the other hand, protons bound to macromolecules, such as proteins, tend to have a fixed orientation and so the average magnetic field in close proximity to such structures does not average to zero. The result is a spatial pattern in the magnetic field that gives rise to a residual dipolar coupling (range of precession frequencies) for the protons experiencing the magnetic field. The wide frequency distribution appears as a broad spectrum that may be several kHz wide. The net signal from these protons disappears very quickly, in inverse proportion to the width, due to the loss of coherence of the spins, i.e. T2 relaxation. Due to exchange mechanisms, such as spin transfer or proton chemical exchange, the (incoherent) spins bound to the macromolecules continually switch places with (coherent) spins in the bulk media and establish a dynamic equilibrium.
Magnetization transfer: Although there is no measurable signal from the bound spins, or the bound spins that exchange into the bulk media, their longitudinal magnetization is preserved and may recover only via the relatively slow process of T1 relaxation. If the longitudinal magnetization of just the bound spins can be altered, then the effect can be measured in the spins of the bulk media due to the exchange processes. The magnetization transfer sequence applies RF saturation at a frequency that is far off resonance for the narrow line of bulk water but still on resonance for the bound protons with a spectral linewidth of kHz. This causes saturation of the bound spins which exchange into the bulk water, resulting in a loss of longitudinal magnetization and hence signal decrease in the bulk water. This provides an indirect measure of macromolecular content in tissue. Implementation of magnetization transfer involves choosing suitable frequency offsets and pulse shapes to saturate the bound spins sufficiently strongly, within the safety limits of specific absorption rate for RF irradiation.
T1ρ (T1rho): Molecules have a kinetic energy that is a function of the temperature and is expressed as translational and rotational motions, and by collisions between molecules. The moving dipoles disturb the magnetic field but are often extremely rapid so that the average effect over a long time-scale may be zero. However, depending on the time-scale, the interactions between the dipoles do not always average away. At the slowest extreme the interaction time is effectively infinite and occurs where there are large, stationary field disturbances (e.g. a metallic implant). In this case the loss of coherence is described as a "static dephasing". T2* is a measure of the loss of coherence in an ensemble of spins that include all interactions (including static dephasing). T2 is a measure of the loss of coherence that excludes static dephasing, using an RF pulse to reverse the slowest types of dipolar interaction. There is in fact a continuum of interaction time-scales in a given biological sample and the properties of the refocusing RF pulse can be tuned to refocus more than just static dephasing. In general, the rate of decay of an ensemble of spins is a function of the interaction times and also the power of the RF pulse. This type of decay, occurring under the influence of RF, is known as T1ρ. It is similar to T2 decay but with some slower dipolar interactions refocused as well as the static interactions.
Fluid attenuated inversion recovery (FLAIR)
Fluid Attenuated Inversion Recovery (FLAIR)[27] is an inversion-recovery pulse sequence used to null signal from fluids. For example, it can be used in brain imaging to suppress cerebrospinal fluid (CSF) so as to bring out the periventricular hyperintense lesions, such as multiple sclerosis (MS) plaques. By carefully choosing the inversion time TI (the time between the inversion and excitation pulses), the signal from any particular tissue can be suppressed.
Magnetic resonance angiography

Magnetic resonance angiography (MRA) generates pictures of the arteries to evaluate them for stenosis (abnormal narrowing) or aneurysms (vessel wall dilatations, at risk of rupture). MRA is often used to evaluate the arteries of the neck and brain, the thoracic and abdominal aorta, the renal arteries, and the legs (called a "run-off"). A variety of techniques can be used to generate the pictures, such as administration of a paramagnetic contrast agent (gadolinium) or using a technique known as "flow-related enhancement" (e.g. 2D and 3D time-of-flight sequences), where most of the signal on an image is due to blood that recently moved into that plane, see also FLASH MRI. Techniques involving phase accumulation (known as phase contrast angiography) can also be used to generate flow velocity maps easily and accurately. Magnetic resonance venography (MRV) is a similar procedure that is used to image veins. In this method, the tissue is now excited inferiorly, while signal is gathered in the plane immediately superior to the excitation plane—thus imaging the venous blood that recently moved from the excited plane.[28]
Magnetic resonance gated intracranial CSF dynamics (MR-GILD)
Magnetic resonance gated intracranial cerebrospinal fluid (CSF) or liquor dynamics (MR-GILD) technique is an MR sequence based on bipolar gradient pulse used to demonstrate CSF pulsatile flow in ventricles, cisterns, aqueduct of Sylvius and entire intracranial CSF pathway. It is a method for analyzing CSF circulatory system dynamics in patients with CSF obstructive lesions such as normal pressure hydrocephalus. It also allows visualization of both arterial and venous pulsatile blood flow in vessels without use of contrast agents.[29][30]
| Diastolic time data acquisition (DTDA). | Systolic time data acquisition (STDA). |
|---|---|
 |
 |
Functional MRI

Functional MRI (fMRI) measures signal
changes in the
brain that are due to changing
neural activity. The brain is scanned at low
resolution but at a rapid rate (typically once
every 2–3 seconds). Increases in neural activity
cause changes in the MR signal via T*
2 changes;[33]
this mechanism is referred to as the BOLD (blood-oxygen-level
dependent) effect. Increased neural activity
causes an increased demand for oxygen, and the
vascular system actually overcompensates for
this, increasing the amount of oxygenated
hemoglobin relative to deoxygenated
hemoglobin. Because deoxygenated hemoglobin
attenuates the MR signal, the vascular response
leads to a signal increase that is related to
the neural activity. The precise nature of the
relationship between neural activity and the
BOLD signal is a subject of current research.
The BOLD effect also allows for the generation
of high resolution 3D maps of the venous
vasculature within neural tissue.
While BOLD signal is the most common method employed for neuroscience studies in human subjects, the flexible nature of MR imaging provides means to sensitize the signal to other aspects of the blood supply. Alternative techniques employ arterial spin labeling (ASL) or weight the MRI signal by cerebral blood flow (CBF) and cerebral blood volume (CBV). The CBV method requires injection of a class of MRI contrast agents that are now in human clinical trials. Because this method has been shown to be far more sensitive than the BOLD technique in preclinical studies, it may potentially expand the role of fMRI in clinical applications. The CBF method provides more quantitative information than the BOLD signal, albeit at a significant loss of detection sensitivity.
Real-time MRI
Real-time MRI refers to the continuous monitoring (“filming”) of moving objects in real time. While many different strategies have been developed over the past two decades, a recent development reported a real-time MRI technique based on radial FLASH that yields a temporal resolution of 20 to 30 milliseconds for images with an in-plane resolution of 1.5 to 2.0 mm. The new method promises to add important information about diseases of the joints and the heart. In many cases MRI examinations may become easier and more comfortable for patients.
Interventional MRI
The lack of harmful effects on the patient and the operator make MRI well-suited for "interventional radiology", where the images produced by a MRI scanner are used to guide minimally invasive procedures. Of course, such procedures must be done without any ferromagnetic instruments.
A specialized growing subset of interventional MRI is that of intraoperative MRI in which the MRI is used in the surgical process. Some specialized MRI systems have been developed that allow imaging concurrent with the surgical procedure. More typical, however, is that the surgical procedure is temporarily interrupted so that MR images can be acquired to verify the success of the procedure or guide subsequent surgical work.
Radiation therapy simulation
Because of MRI's superior imaging of soft tissues, it is now being utilized to specifically locate tumors within the body in preparation for radiation therapy treatments. For therapy simulation, a patient is placed in specific, reproducible, body position and scanned. The MRI system then computes the precise location, shape and orientation of the tumor mass, correcting for any spatial distortion inherent in the system. The patient is then marked or tattooed with points that, when combined with the specific body position, permits precise triangulation for radiation therapy.
Current density imaging
Current density imaging (CDI) endeavors to use the phase information from images to reconstruct current densities within a subject. Current density imaging works because electrical currents generate magnetic fields, which in turn affect the phase of the magnetic dipoles during an imaging sequence.
Magnetic resonance guided focused ultrasound
In MRgFUS therapy, ultrasound beams are focused on a tissue—guided and controlled using MR thermal imaging—and due to the significant energy deposition at the focus, temperature within the tissue rises to more than 65 °C (150 °F), completely destroying it. This technology can achieve precise ablation of diseased tissue. MR imaging provides a three-dimensional view of the target tissue, allowing for precise focusing of ultrasound energy. The MR imaging provides quantitative, real-time, thermal images of the treated area. This allows the physician to ensure that the temperature generated during each cycle of ultrasound energy is sufficient to cause thermal ablation within the desired tissue and if not, to adapt the parameters to ensure effective treatment.
Multinuclear imaging
Hydrogen is the most frequently imaged nucleus in MRI because it is present in biological tissues in great abundance, and because its high gyromagnetic ratio gives a strong signal. However, any nucleus with a net nuclear spin could potentially be imaged with MRI. Such nuclei include helium-3, carbon-13, fluorine-19, oxygen-17, sodium-23, phosphorus-31 and xenon-129. 23Na and 31P are naturally abundant in the body, so can be imaged directly. Gaseous isotopes such as 3He or 129Xe must be hyperpolarized and then inhaled as their nuclear density is too low to yield a useful signal under normal conditions. 17O and 19F can be administered in sufficient quantities in liquid form (e.g. 17O-water) that hyperpolarization is not a necessity.
Multinuclear imaging is primarily a research technique at present. However, potential applications include functional imaging and imaging of organs poorly seen on 1H MRI (e.g. lungs and bones) or as alternative contrast agents. Inhaled hyperpolarized 3He can be used to image the distribution of air spaces within the lungs. Injectable solutions containing 13C or stabilized bubbles of hyperpolarized 129Xe have been studied as contrast agents for angiography and perfusion imaging. 31P can potentially provide information on bone density and structure, as well as functional imaging of the brain.
Susceptibility weighted imaging (SWI)
Susceptibility weighted imaging (SWI), is a new type of contrast in MRI different from spin density, T1, or T2 imaging. This method exploits the susceptibility differences between tissues and uses a fully velocity compensated, three dimensional, RF spoiled, high-resolution, 3D gradient echo scan. This special data acquisition and image processing produces an enhanced contrast magnitude image very sensitive to venous blood, hemorrhage and iron storage. It is used to enhance the detection and diagnosis of tumors, vascular and neurovascular diseases (stroke and hemorrhage, multiple sclerosis, Alzheimer's), and also detects traumatic brain injuries that may not be diagnosed using other methods.
Other specialized MRI techniques
New methods and variants of existing methods
are often published when they are able to
produce better results in specific fields.
Examples of these recent improvements are
T*
2-weighted turbo spin-echo (T2
TSE MRI), double inversion recovery MRI
(DIR-MRI) or phase-sensitive inversion recovery
MRI (PSIR-MRI), all of them able to improve
imaging of the brain lesions.
Another example is MP-RAGE
(magnetization-prepared rapid acquisition with
gradient echo),
which improves images of multiple sclerosis
cortical lesions.
Portable instruments
Portable magnetic resonance instruments are available for use in education and field research. Using the principles of Earth's field NMR, they have no powerful polarizing magnet, so that such instruments can be small and inexpensive. Some can be used for both EFNMR spectroscopy and MRI imaging.[40] The low strength of the Earth's field results in poor signal to noise ratios, requiring long scan times to capture spectroscopic data or build up MRI images.
Research with atomic magnetometers have discussed the possibility for cheap and portable MRI instruments without the large magnet.
MRI versus CT
A computed tomography (CT) scanner uses X-rays, a type of ionizing radiation, to acquire images, making it a good tool for examining tissue composed of elements of a higher atomic number than the tissue surrounding them, such as bone and calcifications (calcium based) within the body (carbon based flesh), or of structures (vessels, bowel). MRI, on the other hand, uses non-ionizing radio frequency (RF) signals to acquire its images and is best suited for soft tissue (although MRI can also be used to acquire images of bones, teeth and even fossils).
Contrast in CT images is generated purely by X-ray attenuation, while a variety of properties may be used to generate contrast in MR images. By variation of scanning parameters, tissue contrast can be altered to enhance different features in an image (see Applications for more details). Both CT and MR images may be enhanced by the use of contrast agents. Contrast agents for CT contain elements of a high atomic number, relative to tissue, such as iodine or barium, while contrast agents for MRI have paramagnetic properties, such as gadolinium and manganese, used to alter tissue relaxation times.
CT and MRI scanners are able to generate multiple two-dimensional cross-sections (slices) of tissue and three-dimensional reconstructions. MRI can generate cross-sectional images in any plane (including oblique planes). In the past, CT was limited to acquiring images in the axial (or near axial) plane. The scans used to be called Computed Axial Tomography scans (CAT scans). However, the development of multi-detector CT scanners with near-isotropic resolution, allows the CT scanner to produce data that can be retrospectively reconstructed in any plane with minimal loss of image quality. For purposes of tumor detection and identification in the brain, MRI is generally superior. However, in the case of solid tumors of the abdomen and chest, CT is often preferred as it suffers less from motion artifacts. Furthermore, CT usually is more widely available, faster, less expensive, and may be less likely to require the person to be sedated or anesthetized[why?].
MRI is also best suited for cases when a patient is to undergo the exam several times successively in the short term, because, unlike CT, it does not expose the patient to the hazards of ionizing radiation.
Economics of MRI
MRI equipment is expensive. 1.5 tesla scanners often cost between US$1 million and US$1.5 million. 3.0 tesla scanners often cost between US$2 million and US$2.3 million. Construction of MRI suites can cost up to US$500,000, or more, depending on project scope.
MRI scanners have been significant sources of revenue for healthcare providers in the US. This is because of favorable reimbursement rates from insurers and federal government programs. Insurance reimbursement is provided in two components, an equipment charge for the actual performance of the MRI scan and professional charge for the radiologist's review of the images and/or data. In the US Northeast, an equipment charge might be $3,500 and a professional charge might be $350 although the actual fees received by the equipment owner and interpreting physician are often significantly less and depend on the rates negotiated with insurance companies or determined by governmental action as in the Medicare Fee Schedule. For example, an orthopedic surgery group in Illinois billed a charge of $1,116 for a knee MRI in 2007 but the Medicare reimbursement in 2007 was only $470.91. Many insurance companies require preapproval of an MRI procedure as a condition for coverage.
In the US, the Deficit Reduction Act of 2005 significantly reduced reimbursement rates paid by federal insurance programs for the equipment component of many scans, shifting the economic landscape. Many private insurers have followed suit.
Safety
A number of features of MRI scanning can give rise to risks.
These include:
- Powerful magnetic fields
- Cryogenic liquids
- Noise
- Claustrophobia
In addition, in cases where MRI contrast agents are used, these also typically have associated risks.
Magnetic field
Most forms of medical or biostimulation implants are generally considered contraindications for MRI scanning. These include pacemakers, vagus nerve stimulators, implantable cardioverter-defibrillators, loop recorders, insulin pumps, cochlear implants, deep brain stimulators and capsules retained from capsule endoscopy. Patients are therefore always asked for complete information about all implants before entering the room for an MRI scan. Several deaths have been reported in patients with pacemakers who have undergone MRI scanning without appropriate precautions.[citation needed] To reduce such risks, implants are increasingly being developed to make them able to be safely scanned, and specialized protocols have been developed to permit the safe scanning of selected implants and pacing devices. Cardiovascular stents are considered safe, however.
Ferromagnetic foreign bodies such as shell fragments, or metallic implants such as surgical prostheses and aneurysm clips are also potential risks. Interaction of the magnetic and radio frequency fields with such objects can lead to trauma due to movement of the object in the magnetic field or thermal injury from radio-frequency induction heating of the object.
Titanium and its alloys are safe from movement from the magnetic field.
In the United States a classification system for implants and ancillary clinical devices has been developed by ASTM International and is now the standard supported by the US Food and Drug Administration:
- MR-Safe — The device or implant is completely non-magnetic, non-electrically conductive, and non-RF reactive, eliminating all of the primary potential threats during an MRI procedure.
- MR-Conditional — A device or implant that may contain magnetic, electrically conductive or RF-reactive components that is safe for operations in proximity to the MRI, provided the conditions for safe operation are defined and observed (such as 'tested safe to 1.5 teslas' or 'safe in magnetic fields below 500 gauss in strength').
- MR-Unsafe — Nearly self-explanatory, this category is reserved for objects that are significantly ferromagnetic and pose a clear and direct threat to persons and equipment within the magnet room.
The very high strength of the magnetic field can also cause "missile-effect" accidents, where ferromagnetic objects are attracted to the center of the magnet, and there have been incidences of injury and death. To reduce the risks of projectile accidents, ferromagnetic objects and devices are typically prohibited in proximity to the MRI scanner and patients undergoing MRI examinations are required to remove all metallic objects, often by changing into a gown or scrubs and ferromagnetic detection devices are used by some sites.
There is no evidence for biological harm from even very powerful static magnetic fields
Radio frequency energy
A powerful radio transmitter is needed for excitation of proton spins. This can heat the body to the point of risk of hyperthermia in patients, particularly in obese patients or those with thermoregulation disorders. Several countries have issued restrictions on the maximum specific absorption rate that a scanner may produce.
|
|
Peripheral nerve stimulation (PNS) |
|
The rapid switching on and off of the magnetic field gradients is capable of causing nerve stimulation. Volunteers report a twitching sensation when exposed to rapidly switched fields, particularly in their extremities. The reason the peripheral nerves are stimulated is that the changing field increases with distance from the center of the gradient coils (which more or less coincides with the center of the magnet). Note however that when imaging the head, the heart is far off-center and induction of even a tiny current into the heart must be avoided at all costs. Although PNS was not a problem for the slow, weak gradients used in the early days of MRI, the strong, rapidly switched gradients used in techniques such as EPI, fMRI, diffusion MRI, etc. are indeed capable of inducing PNS. American and European regulatory agencies insist that manufacturers stay below specified dB/dt limits (dB/dt is the change in field per unit time) or else prove that no PNS is induced for any imaging sequence. As a result of dB/dt limitation, commercial MRI systems cannot use the full rated power of their gradient amplifiers.
|
|
Acoustic noise |
|
Switching of field gradients causes a change in the Lorentz force experienced by the gradient coils, producing minute expansions and contractions of the coil itself. As the switching is typically in the audible frequency range, the resulting vibration produces loud noises (clicking or beeping). This is most marked with high-field machines and rapid-imaging techniques in which sound intensity can reach 120 dB. (equivalent to a jet engine at take-off), and therefore appropriate ear protection is essential for anyone inside the MRI scanner room during the examination.
Noise Reduction Features
General Features
• Acoustically optimized mountings for all
components including gradient coil and body coil
• Minimized structure borne noise transfer to
building
• Noise attenuating foam between gradient coil
and cover, and between magnet and cover
• Encapsulation of noise producing components
Gradient
• Special epoxy resin and casting technology for
damping vibrations
• Reduction of gradient stray field to decrease
eddy currents
• Noise-optimization of the MR system with an
acoustically soft but mechanically rigid
mounting of the gradient coil inside the magnet
• Force compensation for all axes
Magnet
• Encapsulation of the entire magnet
• Efficient floor decoupling for reduction of
noise transferred to the building
• Noise-optimized cold head
Body Coil
Material of supporting tube of the body coil is
optimized for low vibration and noise
• In order to achieve maximum noise reduction,
the body coil tube is extended beyond the
gradient coil
• Copper structures are slotted and glued to the
tube to reduce high frequency noise
• The Body coil is acoustically decoupled by
special suspensions
Sequence Design
Optimized sequence timing.
• Sequences automatically avoid parameter
settings that cause the gradient coil to
resonate
• No relevant application drawbacks – no
increase in sequence parameters, e.g. full
performance
“Whisper Mode”
The “Whisper Mode” is a user selectable mode
that reduces the max. slew rate and max.
amplitude of the gradients and enables very
quiet imaging techniques.
Cryogens
As described in Physics of Magnetic Resonance Imaging, many MRI scanners rely on cryogenic liquids to enable superconducting capabilities of the electromagnetic coils within. Though the cryogenic liquids used are non-toxic, their physical properties present specific hazards.
An unintentional shut-down of a superconducting electromagnet, an event known as "quench", involves the rapid boiling of liquid helium from the device. If the rapidly expanding helium cannot be dissipated through an external vent, sometimes referred to as 'quench pipe', it may be released into the scanner room where it may cause displacement of the oxygen and present a risk of asphyxiation.
Oxygen deficiency monitors are usually used as a safety precaution. Liquid helium, the most commonly used cryogen in MRI, undergoes near explosive expansion as it changes from liquid to a gaseous state. The use of an oxygen monitor is important for two reasons: 1. Keeping the oxygen levels safe for patient/physicians and; 2. Making sure the MRI Scanner is working properly. Rooms built in support of superconducting MRI equipment should be equipped with pressure relief mechanisms and an exhaust fan, in addition to the required quench pipe.
Since a quench results in rapid loss of all cryogens in the magnet, recommissioning the magnet is expensive and time-consuming. Spontaneous quenches are uncommon, but may also be triggered by equipment malfunction, improper cryogen fill technique, contaminants inside the cryostat, or extreme magnetic or vibrational disturbances.
Contrast agents
The most commonly used intravenous contrast agents are based on chelates of gadolinium. In general, these agents have proved safer than the iodinated contrast agents used in X-ray radiography or CT. Anaphylactoid reactions are rare, occurring in approx. 0.03–0.1%.[61] Of particular interest is the lower incidence of nephrotoxicity, compared with iodinated agents, when given at usual doses—this has made contrast-enhanced MRI scanning an option for patients with renal impairment, who would otherwise not be able to undergo contrast-enhanced CT.
Although gadolinium agents have proved useful for patients with renal impairment, in patients with severe renal failure requiring dialysis there is a risk of a rare but serious illness, nephrogenic systemic fibrosis, that may be linked to the use of certain gadolinium-containing agents. The most frequently linked is gadodiamide, but other agents have been linked too. Although a causal link has not been definitively established, current guidelines in the United States are that dialysis patients should only receive gadolinium agents where essential, and that dialysis should be performed as soon as possible after the scan to remove the agent from the body promptly. In Europe, where more gadolinium-containing agents are available, a classification of agents according to potential risks has been released. Recently a new contrast agent named gadoxetate, brand name Eovist (US) or Primovist (EU), was approved for diagnostic use: this has the theoretical benefit of a dual excretion path.
Pregnancy
No effects of MRI on the fetus have been demonstrated. In particular, MRI avoids the use of ionizing radiation, to which the fetus is particularly sensitive. However, as a precaution, current guidelines recommend that pregnant women undergo MRI only when essential. This is particularly the case during the first trimester of pregnancy, as organogenesis takes place during this period. The concerns in pregnancy are the same as for MRI in general, but the fetus may be more sensitive to the effects—particularly to heating and to noise. However, one additional concern is the use of contrast agents; gadolinium compounds are known to cross the placenta and enter the fetal bloodstream, and it is recommended that their use be avoided.
Despite these concerns, MRI is rapidly growing in importance as a way of diagnosing and monitoring congenital defects of the fetus because it can provide more diagnostic information than ultrasound and it lacks the ionizing radiation of CT. MRI without contrast agents is the imaging mode of choice for pre-surgical, in-utero diagnosis and evaluation of fetal tumors, primarily teratomas, facilitating open fetal surgery, other fetal interventions, and planning for procedures (such as the EXIT procedure) to safely deliver and treat babies whose defects would otherwise be fatal.
Claustrophobia and discomfort
Due to the construction of some MRI scanners, they can be potentially unpleasant to lie in. Older models of closed bore MRI systems feature a fairly long tube or tunnel. The part of the body being imaged must lie at the center of the magnet, which is at the absolute center of the tunnel. Because scan times on these older scanners may be long (occasionally up to 40 minutes for the entire procedure), people with even mild claustrophobia are sometimes unable to tolerate an MRI scan without management. Modern scanners may have larger bores (up to 70 cm) and scan times are shorter. This means that claustrophobia is less of an issue, and many patients now find MRI an innocuous and easily tolerated procedure.
Nervous patients may still find the following strategies helpful:
- Advance preparation
- visiting the scanner to see the room and practice lying on the table
- visualization techniques
- chemical sedation
- general anesthesia
- Coping while inside the scanner
- holding a "panic button"
- closing eyes as well as covering them (e.g. washcloth, eye mask)
- listening to music on headphones or watching a movie, using mirror-glasses and a projection screen or via a Head-mounted display, while in the machine.
Alternative scanner designs, such as open or upright systems, can also be helpful where these are available. Though open scanners have increased in popularity, they produce inferior scan quality because they operate at lower magnetic fields than closed scanners. However, commercial 1.5 tesla open systems have recently become available, providing much better image quality than previous lower field strength open models.
For babies and young children chemical sedation or general anesthesia are the norm, as these subjects cannot be instructed to hold still during the scanning session. Obese patients and pregnant women may find the MRI machine to be a tight fit. Pregnant women may also have difficulty lying on their backs for an hour or more without moving.
Guidance
Safety issues, including the potential for
biostimulation device interference, movement of
ferromagnetic bodies, and incidental localized
heating, have been addressed in the
American College of Radiology's White
Paper on MR Safety, which was originally
published in 2002 and expanded in 2004. The
ACR White Paper on MR Safety has been
rewritten and was released early in 2007 under
the new title
ACR Guidance Document for Safe MR Practices.
In December 2007, the Medicines in Healthcare
product Regulation Agency (MHRA), a UK
healthcare regulatory body, issued their
Safety Guidelines for Magnetic Resonance
Imaging Equipment in Clinical Use.
In February 2008, the
Joint Commission, a US healthcare
accrediting organization, issued a
Sentinel Event Alert #38, their highest
patient safety advisory, on MRI safety issues.
In July 2008, the United States Veterans
Administration, a federal governmental agency
serving the healthcare needs of former military
personnel, issued a substantial revision to
their
MRI Design Guide, which includes
physical or facility safety considerations.
The European Physical Agents Directive
The European Physical Agents (Electromagnetic Fields) Directive is legislation adopted in European legislature. Originally scheduled to be required by the end of 2008, each individual state within the European Union must include this directive in its own law by the end of 2012. Some member nations passed complying legislation and are now attempting to repeal their state laws in expectation that the final version of the EU Physical Agents Directive will be substantially revised prior to the revised adoption date.
The directive applies to occupational exposure to electromagnetic fields (not medical exposure) and was intended to limit workers’ acute exposure to strong electromagnetic fields, as may be found near electricity substations, radio or television transmitters or industrial equipment. However, the regulations impact significantly on MRI, with separate sections of the regulations limiting exposure to static magnetic fields, changing magnetic fields and radio frequency energy. Field strength limits are given, which may not be exceeded. An employer may commit a criminal offense by allowing a worker to exceed an exposure limit, if that is how the Directive is implemented in a particular member state.
The Directive is based on the international consensus of established effects of exposure to electromagnetic fields, and in particular the advice of the European Commissions's advisor, the International Commission on Non-Ionizing Radiation Protection (ICNIRP). The aims of the Directive, and the ICNIRP guidelines it is based on, are to prevent exposure to potentially harmful fields. The actual limits in the Directive are very similar to the limits advised by the Institute of Electrical and Electronics Engineers, with the exception of the frequencies produced by the gradient coils, where the IEEE limits are significantly higher.
Many Member States of the EU already have either specific EMF regulations or (as in the UK) a general requirement under workplace health and safety legislation to protect workers against electromagnetic fields. In almost all cases the existing regulations are aligned with the ICNIRP limits so that the Directive should, in theory, have little impact on any employer already meeting their legal responsibilities.
The introduction of the Directive has brought to light an existing potential issue with occupational exposures to MRI fields. There are at present very few data on the number or types of MRI practice that might lead to exposures in excess of the levels of the Directive. There is a justifiable concern amongst MRI practitioners that if the Directive were to be enforced more vigorously than existing legislation, the use of MRI might be restricted, or working practices of MRI personnel might have to change.
In the initial draft a limit of static field strength to 2 T was given. This has since been removed from the regulations, and whilst it is unlikely to be restored as it was without a strong justification, some restriction on static fields may be reintroduced after the matter has been considered more fully by ICNIRP. The effect of such a limit might be to restrict the installation, operation and maintenance of MRI scanners with magnets of 2 T and stronger. As the increase in field strength has been instrumental in developing higher resolution and higher performance scanners, this would be a significant step back. This is why it is unlikely to happen without strong justification.
Individual government agencies and the European Commission have now formed a working group to examine the implications on MRI and to try to address the issue of occupational exposures to electromagnetic fields from MRI.
Three-dimensional (3D) image reconstruction
The principle
Because contemporary MRI scanners offer isotropic, or near isotropic, resolution, display of images does not need to be restricted to the conventional axial images. Instead, it is possible for a software program to build a volume by 'stacking' the individual slices one on top of the other. The program may then display the volume in an alternative manner.
3D rendering techniques
- Surface rendering
- A threshold value of greyscale density is chosen by the operator (e.g. a level that corresponds to fat). A threshold level is set, using edge detection image processing algorithms. From this, a 3-dimensional model can be constructed and displayed on screen. Multiple models can be constructed from various different thresholds, allowing different colors to represent each anatomical component such as bone, muscle, and cartilage. However, the interior structure of each element is not visible in this mode of operation.
- Volume rendering
- Surface rendering is limited in that it only displays surfaces that meet a threshold density, and only displays the surface closest to the imaginary viewer. In volume rendering, transparency and colors are used to allow a better representation of the volume to be shown in a single image - e.g. the bones of the pelvis could be displayed as semi-transparent, so that even at an oblique angle, one part of the image does not conceal another.
Image segmentation
Where different structures have similar threshold density, it can become impossible to separate them simply by adjusting volume rendering parameters. The solution is called segmentation, a manual or automatic procedure that can remove the unwanted structures from the image.